Please select a subject first
Advertisements
Advertisements
The following solutions were prepared by dissolving 10 g of glucose (C6H12O6) in 250 ml of water (P1), 10 g of urea (CH4N2O) in 250 ml of water (P2) and 10 g of sucrose (C12H22O11) in 250 ml of water (P3). The right option for the decreasing order of osmotic pressure of these solutions is
Concept: undefined >> undefined
Match List - I with List - II.
| List - I | List - II | ||
| (a) | 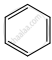 \[\ce{->[CO,HCl][Anhyd. AlCl3/CuCl]}\] \[\ce{->[CO,HCl][Anhyd. AlCl3/CuCl]}\] |
(i) | Hell-Volhard-Zelinsky reaction |
| (b) |
\[\begin{array}{cc}
\ce{O}\phantom{.................}\\ ||\phantom{.................}\\ \ce{R - C - CH3 + NaOX ->} \end{array}\] |
(ii) | Gattermann-Koch reaction |
| (c) | \[\ce{R - CH2 - OH + R'COOH ->[Conc. H2SO4]}\] | (iii) | Haloform reaction |
| (d) | \[\ce{R - CH2COOH ->[(i) X2/Red P][(ii) H2O]}\] | (iv) | Esterification |
Choose the correct answer from the options given below.
Concept: undefined >> undefined
Advertisements
Which of the following vitamins is water soluble?
Concept: undefined >> undefined
One part of an element A combines with two parts of an element B. Six parts of the element C combines with four parts of element B. If A and C combine together the ratio of their weights will be governed by ______.
Concept: undefined >> undefined
Choose the INCORRECT statement.
Concept: undefined >> undefined
In a period, element with least atomic size is ______.
Concept: undefined >> undefined
Which of the following acids is a vitamin?
Concept: undefined >> undefined
The value of ΔH for the reaction, \[\ce{X2_{(g)} + 4Y2_{(g)} ⇌ 2XY4_{(g)}}\] is less than zero. Formation of \[\ce{XY4_{(g)}}\] will be favoured at ______.
Concept: undefined >> undefined
Which one of the following statements about the zeolites is CORRECT?
Concept: undefined >> undefined
The reaction of chloroform with Ag powder gives ______.
Concept: undefined >> undefined
At CMC (Critical micelle concentration) the surface molecules ______.
Concept: undefined >> undefined
Uncertainty principle was given by ______.
Concept: undefined >> undefined
The number of dative bonds in sulphric acid molecule is ______.
Concept: undefined >> undefined
Siderite is a mineral of ______
Concept: undefined >> undefined
What is the catalyst used for the oxidation of SO2 to SO3 in the lead chamber process for the manufacture of sulphuric acid?
Concept: undefined >> undefined
Indicate the wrong statement according to Valence bond theory:
Concept: undefined >> undefined
The ratio of root mean square speed, average speed and most probable speed is ______.
Concept: undefined >> undefined
The reaction given below \[\ce{BaCl2(g) + 2H2O(ℓ) -> BaCl2 . 2H2O(s);}\] ΔH = – x KJ. The value of ΔH represents:
Concept: undefined >> undefined
Action of zinc on tetrabromoethane gives:
Concept: undefined >> undefined
Benzenediazonium chloride reacts with phenol to give p-hydroxy azobenzene, an orange dye. This reaction is known as ______.
Concept: undefined >> undefined
Advertisements
Advertisements
| National Testing Agency NEET (UG) Medical Entrance Exam Question Bank Solutions |
|---|
| Question Bank Solutions for National Testing Agency NEET (UG) Medical Entrance Exam Biology |
| Question Bank Solutions for National Testing Agency NEET (UG) Medical Entrance Exam Chemistry |
| Question Bank Solutions for National Testing Agency NEET (UG) Medical Entrance Exam Physics |
