Please select a subject first
Advertisements
Advertisements
Balance the following reaction by oxidation number method.
\[\ce{Cr2O^2-_{7(aq)} + SO^2-_{3(aq)}->Cr^3+_{ (aq)} + SO^2-_{4(aq)}(acidic)}\]
Concept: undefined >> undefined
Balance the following reaction by oxidation number method.
\[\ce{MnO^-_{4(aq)} + Br^-_{ (aq)}->MnO2_{ (s)} + BrO^-_{3(aq)}(basic)}\]
Concept: undefined >> undefined
Advertisements
Balance the following reaction by oxidation number method.
\[\ce{H2SO4_{(aq)} + C_{(s)} -> CO2_{(g)} + SO2_{(g)} + H2O_{(l)}(acidic)}\]
Concept: undefined >> undefined
Balance the following reaction by oxidation number method.
\[\ce{Bi(OH)_{3(s)} + Sn(OH)^-_{3(aq)}->Bi_{(s)} + Sn(OH)^2-_{6(aq)}(basic)}\]
Concept: undefined >> undefined
Balance the following redox equation by half-reaction method.
\[\ce{H2C2O_{4(aq)} + MnO^-_{4(aq)}->CO2_{(g)} + Mn^2+_{( aq)}(acidic)}\]
Concept: undefined >> undefined
Balance the following redox equation by half-reaction method.
\[\ce{Bi(OH)_{3(s)} + SnO^2-_{2(aq)}->SnO^2-_{3(aq)} + Bi^_{(s)}(basic)}\]
Concept: undefined >> undefined
Write the outer electronic configuration of the following using the orbital notation method. Justify.
Ge (belongs to period 4 and group 14)
Concept: undefined >> undefined
Write the outer electronic configuration of the following using the orbital notation method. Justify.
Po (belongs to period 6 and group 16)
Concept: undefined >> undefined
Write the outer electronic configuration of the following using the orbital notation method. Justify.
Cu (belongs to period 4 and group 11)
Concept: undefined >> undefined
Answer the following.
La belongs to group 3 while Hg belongs to group 12 and both belong to period 6 of the periodic table. Write down the general outer electronic configuration of the ten elements from La to Hg together using the orbital notation method.
Concept: undefined >> undefined
Find out all the functional groups present in the following polyfunctional compound.
Thyroxine, the principal thyroid hormone.

Concept: undefined >> undefined
Find out all the functional groups present in the following polyfunctional compound.
Penicillin G, a naturally occurring antibiotic.
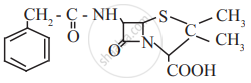
Concept: undefined >> undefined
Identify primary, secondary, tertiary, and quaternary carbon in the following compound.
\[\begin{array}{cc}\ce{CH3}\phantom{..................}\\
|\phantom{....................}\\\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\|\phantom{.....}|\phantom{................}\\
\ce{CH3}\phantom{.}\ce{CH3}\phantom{..............}
\end{array}\]
Concept: undefined >> undefined
Identify primary, secondary, tertiary and quaternary carbon in the following compound.
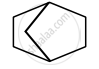
Concept: undefined >> undefined
Answer the following question.
The electronic configuration of some element is given below:
1s2
In which group and period of the periodic table the element is placed?
Concept: undefined >> undefined
Answer the following question.
The electronic configuration of some element is given below:
1s2 2s2 2p6
In which group and period of the periodic table the element is placed?
Concept: undefined >> undefined
What is meant by homologous series?
Concept: undefined >> undefined
Write the first four members of the homologous series that begins with CH3CHO. Also, write down their general molecular formula.
Concept: undefined >> undefined
Write the first four members of the homologous series that begins with H-C≡C-H. Also, write down their general molecular formula.
Concept: undefined >> undefined
Answer the following question.
For s-block and p-block elements show that the number of valence electrons is equal to its group number.
Concept: undefined >> undefined
