Advertisements
Advertisements
The heats of atomisation of PH3(g) and P2H4(g) are 954 kJ mol−1 and 1485 kJ mol respectively. The P-P bond energy in kJ mol−1 is ____________.
Concept: undefined >> undefined
The below given molecular geometry corresponds to which of the following molecule?
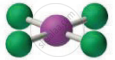
Concept: undefined >> undefined
Advertisements
Identify the INCORRECT statement.
Concept: undefined >> undefined
Which of the following statements are FALSE about lanthanoids?
(I) They have lower heat of atomization than transition metals
(II) They are soft metals with silvery white colour and densities of about 7 g cm−3.
(III) They react with water to give the metal hydroxide and oxygen gas.
(IV) They form hydroxides, which are covalent and basic.
Concept: undefined >> undefined
Which mineral among the following contains vanadium in it?
Concept: undefined >> undefined
Which of the following is CORRECT about alkynes?
Concept: undefined >> undefined
The designation of a subshell with n = 6 and l = 2 is ____________.
Concept: undefined >> undefined
The electronic configuration of transition element 'X' in +3 oxidation state is [Ar] 3d4. What is its atomic number?
Concept: undefined >> undefined
Which among the following is used in the treatment of cancer?
Concept: undefined >> undefined
For a process, entropy change of a system is expressed as ________.
Concept: undefined >> undefined
For a reversible spontaneous change, ∆S is ____________.
Concept: undefined >> undefined
Which one of the following is an organometallic compound?
Concept: undefined >> undefined
Which one of the following orders is CORRECT in case of energy of the given subshells?
P: n = 4; l = 3
Q: n = 5; I = 1
R: n = 5; l = 0
S: n = 4; l = 2
Concept: undefined >> undefined
Arrange the following in order of increasing entropy.
I. 1.0 mol H2O (298 K, 1 atm)
II. 1.0 mol H2O (270 K, 1 atm)
III. 1.0 mol H2O (400 K, 1 atm)
Concept: undefined >> undefined
How many electrons in 19K have n = 3, l = 1?
Concept: undefined >> undefined
The hybridization of the central atom in BF3, CH4, NH3 and BeCl2 are ____________ respectively.
Concept: undefined >> undefined
Which statement(s) is/are CORRECT about applications of lanthanoid metals/ions and their compounds?
(I) The optoelectronics applications use lanthanoid ions as active ions in luminescent materials.
(II) Lanthanoid compounds are used in television tubes.
(III) Lanthanoids are used in hybrid cars, superconductors and permanent magnets.
Concept: undefined >> undefined
Which of the following alcohol contains −OH group attached to a sp2 hybridised carbon atom?
Concept: undefined >> undefined
The reaction in which 2 molecules of chlorobenzene reacts with metallic sodium in presence of dry ether forming diphenyl is an example of ______.
Concept: undefined >> undefined
How many gram of sodium (atomic mass 23 u) is required to prepare one mole of ethane from methyl chloride by Wurtz reaction?
Concept: undefined >> undefined
