Advertisements
Advertisements
Question
The below given molecular geometry corresponds to which of the following molecule?
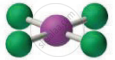
Options
SiCl4
SF4
XeF4
CH4
MCQ
Advertisements
Solution
XeF4
Explanation:
The molecular geometry is square planar in this case. The only XeF4 is the square planar of the other alternatives.
SiCl4 and CH4 have a tetrahedral shape, but SF4 has a see-saw shape.
shaalaa.com
Hybridization
Is there an error in this question or solution?
