Please select a subject first
Advertisements
Advertisements
Lanthanoids and actinoids resemble in ______.
Concept: undefined >> undefined
Identify the weakest oxidising agent among the following.
Concept: undefined >> undefined
Advertisements
Nitration ofwhich among the following compounds yields cyclonite?
Concept: undefined >> undefined
The Wurtz-Fittig reaction involves condensation of ______.
Concept: undefined >> undefined
Predict the product C obtained in the following reaction of butyne-1.
\[\ce{CH3CH2 - C ≡ CH + HCl -> B ->[HI] C}\]
Concept: undefined >> undefined
Standard enthalpy and standard entropy changes for the oxidation of ammonia at 298 K are −382.64 kJ mol−1 and −145.6 JK−1 mol−1, respectively. Standard Gibb's energy change for the same reaction at 298 K is ______.
Concept: undefined >> undefined
Which of the following reaction is used to prepare toluene from bromobenzene?
Concept: undefined >> undefined
Which of the following factors may be regarded as the main cause of lanthanoid contraction?
Concept: undefined >> undefined
Which of the following is called Wilkinson's catalyst?
Concept: undefined >> undefined
Which of the following cannot be considered as an application of electrochemical series?
Concept: undefined >> undefined
The most basic hydroxide from the following is:
Concept: undefined >> undefined
Calculate the value `"E"_"cell"^circ` at 298 K for the following cell.
\[\ce{Al | Al^{3+} (0.01 M) || Sn^{2+} (0.015 M) | Sn}\]
`"E"_("Al"^(3+)//"Al")^circ` = - 1.66 V and `"E"_("Sn"^(2+)//"Sn")^circ` = - 0.14 V
Concept: undefined >> undefined
In which of the following reaction the value of Kp will be equal to Kc?
Concept: undefined >> undefined
Dinitrogen and dioxygen combined to form nitrous oxide in equilibrium at 850 K. The equilibrium constant for this reaction is 0.56. If the equilibrium concentration of nitrous oxide gas is 3 × 10-3 M and both dinitrogen and dioxygen have the same concentration, then what will be the concentration of dinitrogen gas in M?
Concept: undefined >> undefined
Identify the last element in lanthanoid series.
Concept: undefined >> undefined
Identify the product obtained in the following reaction.
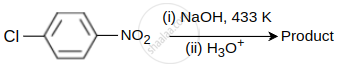
Concept: undefined >> undefined
The major product obtained in the following reaction is
\[\ce{Chlorobenzene + Chlorine ->[Anhydrous][FeCl3] product (Major)}\]
Concept: undefined >> undefined
Identify electron withdrawing group from following when attached to π - bond.
Concept: undefined >> undefined
Which of the following alkyl halide is treated with sodium metal to obtain 2, 2, 3, 3 tetramethyl butane?
Concept: undefined >> undefined
Consider the following sequence of reactions
\[\ce{CH ≡ CH ->[HBr] A ->[HBr] B ->[Alc. KOH]C->[NaNH2]D}\]
Identify D.
Concept: undefined >> undefined
