Please select a subject first
Advertisements
Advertisements
____________ is NOT a simple protein.
Concept: undefined >> undefined
Nucleotide differs from a nucleoside as it has ______
Concept: undefined >> undefined
Advertisements
Choose the WRONG statement from the following.
Concept: undefined >> undefined
The following compound is a/an ____________.
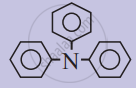
Concept: undefined >> undefined
Which of the following is NOT an actinoid?
Concept: undefined >> undefined
In the reaction,
\[\ce{CH3COOH ->[SOCl2] X ->[Sodium salt of carboxylic acid] Y}\].
The compound Y was found to be a mixed acid anhydride. Thus, the sodium salt of carboxylic acid used CANNOT be ____________.
Concept: undefined >> undefined
The optically inactive amino acid is ____________.
Concept: undefined >> undefined
The conjugate acid of \[\ce{CO^2-_3}\] is ____________.
Concept: undefined >> undefined
At constant temperature, a quantity of an ideal gas occupies 50 mL at 500 mmHg pressure. At what pressure, the volume will be 100 mL?
Concept: undefined >> undefined
Identify the enzyme X in the following reaction.
\[\ce{H2O2(aq) ->[x] H2O(l) + \frac{1}{2} O2(g)}\]
Concept: undefined >> undefined
Identify the element having highest enthalpy of atomisation from following.
Concept: undefined >> undefined
Which among the following lanthanoids, shows only +3 oxidation state?
Concept: undefined >> undefined
Which of the following compounds reacts with ammonia to form urotropine?
Concept: undefined >> undefined
Identify the enzyme that catalyses the reaction of CO2 with water in huma body.
Concept: undefined >> undefined
Which among the following amino acids has lowest molar mass?
Concept: undefined >> undefined
Which of the following is a lanthanoid?
Concept: undefined >> undefined
Find INCORRECT statement with reference to NICAD cell.
Concept: undefined >> undefined
A cold drink bottle contains 200 mL liquid, in which CO2 is 0.1 molar. Considering CO2 as an ideal gas the volume of the dissolved CO2 at S.T.P is ______.
Concept: undefined >> undefined
The bacteriostatic antibiotic from the following is ______.
Concept: undefined >> undefined
In the reaction, \[\ce{C6H5COCH3 ->[{[H]}][Zn-Hg/conc. HCl] X}\], X is ______.
Concept: undefined >> undefined
