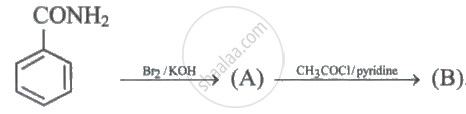
Please select a subject first
Advertisements
Advertisements
Identify the CORRECT statement.
Concept: undefined >> undefined
In phenols, −OH group is attached to ___________ hybridised carbon.
Concept: undefined >> undefined
Advertisements
Which of the following is CORRECT?
\[\ce{H3PO4_{(aq)} + 3KOH_{(aq)} -> K3PO4_{(aq)} + 3H2O_{(l)}}\]
Concept: undefined >> undefined
At 25°C and 1 atm, a cylinder containing 10 L of an ideal gas is connected to the empty cylinder with a capacity of 20 L. The pressures exerted by gas m both the cylinders will be ____________.
Concept: undefined >> undefined
Identify the product obtained when benzyl chloride undergoes ammonolysis in presence of excess ammonia followed by the reaction with two moles of methyl iodide.
Concept: undefined >> undefined
Alkyl halides can be hydrolysed to alcohols by reacting with ____________.
Concept: undefined >> undefined
In the reaction,
\[\ce{MnO^{-1}_4 (aq) + Br^{-1}(aq) -> MnO2(s) + BrO^{-1}_3(aq)}\]
the correct change in oxidation number of the species involved is ______.
Concept: undefined >> undefined
The slope and intercept for plot of `log_10 "x"/"m"` against log10P for Freundlich equation are ____________ and ____________ respectively.
Concept: undefined >> undefined
Which among the following compounds is used as selective weed killer?
Concept: undefined >> undefined
Which of the following represents Etard's reaction?
Concept: undefined >> undefined
α-butylene when subjected to hydroboration oxidation reaction, yields ______.
Concept: undefined >> undefined
Which of the following is NOT an example of redox reaction?
Concept: undefined >> undefined
Phenol is obtained from cumene ____________.
Concept: undefined >> undefined
Which property of an element is always a whole number?
Concept: undefined >> undefined
Which of the following amines exhibits maximum degree of intermolecular hydrogen bonding?
Concept: undefined >> undefined
In the complex [Co(en)3]Cl3, ____________.
Concept: undefined >> undefined
Alkyl fluorides are prepared by heating alkyl bromides or chlorides in the presence of metallic fluorides. The reaction is known as ____________ reaction.
Concept: undefined >> undefined
In the following reaction, the oxidation number of Cr changes.
\[\ce{ClO^-_{( aq)} + Cr(OH)^-_{4(aq)} -> CrO^{2-}_{4(aq)} + Cl^-_{( aq)} (basic)}\]
Concept: undefined >> undefined
In a closed insulated container, a liquid is stirred with a paddle to increase its temperature. In this process, which of the following is TRUE?
Concept: undefined >> undefined