Advertisements
Advertisements
4.0 moles of argon and 5.0 moles of PCl5 are introduced into an evacuated flask of 100 litre capacity at 610 K. The system is allowed to equilibrate. At equilibrium, the total pressure of mixture was found to be 6.0 atm. The Kp for the reaction is ______. [Given: R = 0.082 L atm K-1 mol-1]
Concept: undefined >> undefined
A 42.12% (w/v) solution of NaCl causes precipitation of a certain sol in 10 hours. The coagulating value of NaCl for the sol is ______.
[Given: Molar Mass : Na = 23.0 g mol-1; Cl = 35.5 g mol-1]
Concept: undefined >> undefined
Advertisements
Match List-I with List-II.
| List-I | List-II |
| (Metal) | (Emitted light wavelength (nm)) |
| (a) Li | (I) 780.0 |
| (b) Na | (II) 455.5 |
| (c) Rb | (III) 670.8 |
| (d) Cs | (IV) 589.2 |
Choose the most appropriate answer from the options given below:
Concept: undefined >> undefined
Assertion (A): The first ionization enthalpy for oxygen is lower than that of nitrogen.
Reason (R): The four electrons in 2p orbitals of oxygen experience more electron-electron repulsion.
In light of the above statements, choose the correct answer from the options given below.
Concept: undefined >> undefined
Match List-I with List-II.
| List-I | List-II |
| (Metal) | (Application) |
| (a) Cs | (I) High temperature thermometer |
| (b) Ga | (II) Water repellent sprays |
| (c) B | (III) Photoelectric cells |
| (d) Si | (IV) Bullet proof vest |
Choose the most appropriate answer from the options given below:
Concept: undefined >> undefined
A white precipitate was formed when BaCl2 was added to extract of an inorganic salt. Farther, a gas 'X' with characteristic odour was released when the formed white precipitate was dissolved in dilute HCl. The anion present in the inorganic salt is ______.
Concept: undefined >> undefined
Which is low spin complex?
Concept: undefined >> undefined
The major components in "Gun Metal" are ______.
Concept: undefined >> undefined
\[\ce{2SO2 (g) + O2 (g) <=> 2SO3 (g)}\]
In an equilibrium mixture, the partial pressures are
`"P"_("SO"_2)` = 43 kPa; `"P"_("O"_2)` = 530 Pa and
`"P"_("SO"_2)` = 45 kPA. The equilibrium constant Kp = _____ × 10-2. (Nearest integer)
Concept: undefined >> undefined
Two salts A2X and MX have the same value of solubility product of 4.0 × 10−12. The ratio of their molar solubilities i.e., `("S"("A"_2"X"))/("S"("MX")` = ______. (Round off to the Nearest Integer)
Concept: undefined >> undefined
The metal mainly used in devising photoelectric cells is ______.
Concept: undefined >> undefined
The correct order of hydration enthapies of alkali metal ions is ______.
Concept: undefined >> undefined
The major product of the following reaction is:
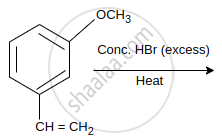
Concept: undefined >> undefined
The major product of the following reaction is:
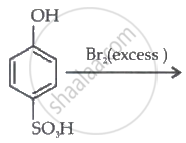
Concept: undefined >> undefined
\[\ce{CH ≡ CH ->[NH4Cl][Cu2Cl2] Product}\]
Product is:
Concept: undefined >> undefined
Nitrogen combines with metals to form ______.
Concept: undefined >> undefined
Which of the following is a double salt?
Concept: undefined >> undefined
The major product of the following reaction is:
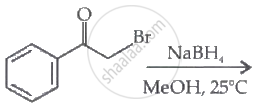
Concept: undefined >> undefined
The arrangement of oxygen atoms around phosphorus atoms in P4O10 is ______.
Concept: undefined >> undefined
Lassaigne's test for the detection of nitrogen fails in ______.
Concept: undefined >> undefined
