Advertisements
Advertisements
Which of the following compounds are formed when boron trichloride is treated with water?
Concept: undefined >> undefined
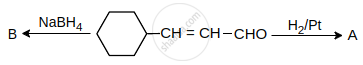
A and B are:
Concept: undefined >> undefined
Advertisements
A pyranose ring consists of a skeleton of ______.
Concept: undefined >> undefined
A mixture of benzene and chloroform is separated by ______.
Concept: undefined >> undefined
MgBr2 and Mgl2 are soluble in acetone because of ______.
Concept: undefined >> undefined
On the addition of mineral acid to an aqueous solution of borax, the compound formed is ______.
Concept: undefined >> undefined
\[\ce{X <-[red P][HI] CH3COOH ->[LiAlH4] Y.}\]
What does Not true for X and Y?
Concept: undefined >> undefined
 |
|
| Fig. I | Fig. II |
In figure-I, an air column of length ℓ1 is entrapped by a column of Hg of length 8 cm. In figure-II length of same air column at the same temperature is ℓ2. The `ℓ_1/ℓ_2` is:
(1 atm = 76 cm of Hg)
Concept: undefined >> undefined
Boron reacts with nitric acid to form ______.
Concept: undefined >> undefined
When aqueous solution of AICl3 is concentrated, it furnishes crystals of ______.
Concept: undefined >> undefined
2 litre He gas at 2 atm and 300 K is inserted into a 4 litre rigid container containing N2 at 600 K and 4 atm. Finally mixture is maintained at 600 K temperature. The final pressure of gaseous mixture would be ______ torr.
Concept: undefined >> undefined
Two gases A and B having molecular weights 60 and 45 respectively are enclosed in a vessel. The wt. of A is 0.50 g and that of B is 0.2 g. The total pressure of the mixture is 750 mm. The partial pressure of the gases B is ______ mm Hg.
Concept: undefined >> undefined
Which one of the following methods is used to prepare borax crystals?
Concept: undefined >> undefined
PbCl4 exists but PbBr4 and Pbl4 do not because of ______.
Concept: undefined >> undefined
In the given reaction final product(s) will be:
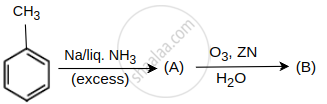
Concept: undefined >> undefined
A gaseous mixture of He and O2 is found to have a density of 0.518 gL-1 at 25° C and 720 torr. The mass percent of helium in this mixture is ______.
Concept: undefined >> undefined
The oxidation state of manganese in the product obtained in a reaction of potassium permanganate and hydrogen peroxide in a basic medium is ______.
Concept: undefined >> undefined
Potassium permanganate on heating at 513 K gives a product which is ______.
Concept: undefined >> undefined
Potassium dichromate when heated with concentrated sulphuric acid and a soluble chloride, gives brown-red vapours of ______.
Concept: undefined >> undefined

