Advertisements
Advertisements
For a chemical reaction starting with some initial concentration of reactant At as a function of time (t) is given by the equation,
`1/("A"_"t"^4) = 2 + 1.5 xx 10^-3` t
The rate of disappearance of [A] is ____ × 10-2 M/sec when [A] = 2 M.
[Given: [At] in M and t in sec.]
[Express your answer in terms of 10-2 M /s]
[Round off your answer if required]
Concept: undefined >> undefined
A strong current of trivalent gaseous boron passed through a silicon crystal decreases the density of the crystal due to part replacement of silicon by boron and due to interstitial vacancies created by missing Si atoms. In one such experiment, one gram of silicon is taken and the boron atoms are found to be 1000 ppm by weight, when the density of the Si crystal decreases by 12%. The percentage of missing vacancies due to silicon, which are filled up by boron atoms will be ____ g. [Given · Atomic wt. Si= 30, B = 11]
Concept: undefined >> undefined
Advertisements
Consider an isothermal cylinder and massless piston assembly in which ideal gas is filled. Cross-sectional area of the cylinder = 1 m2. Three masses m1, m2 and m3 are kept on the piston. When m1 is removed, the piston moves upto point A. When m1 and m2 both are removed piston moves upto point B and when m1, m2 and m3 all three are removed, piston moves upto point C. The work done by the gas is ______ J, when piston moves from point B to point C.
[Given : m1 = 2 × 104 kg, m2 = 3 × 104 kg, g = 10 m/s2]
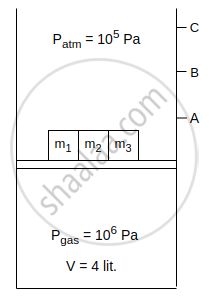
Concept: undefined >> undefined
The density of solid Argon is 1.6 ml at - 233°C. If the Argon atom is assumed to be sphere of radius 1.5 × 10-8 cm, then the ______ % of solid Argon is apparently occupied.
[Take : NA = 6 × 1023, Atomic mass of Ar = 40]
Concept: undefined >> undefined
On small scale, boron is best isolated by reducing boron trioxide with ______.
Concept: undefined >> undefined
A given solution of H2O2 is 30 volumes. Its concentration in terms of molarity is ______.
Concept: undefined >> undefined
Glycinato ligand is ______.
Concept: undefined >> undefined
The composition of a sample of wustite is Fe0.93O1.0· The iron is present in the form of Fe(III) is ______%.
Concept: undefined >> undefined
A sample of Ferrous sulphide reacts with dil. H2SO4 to from H2S which contains 9% hydrogen by volume. The percentage of fee in the sample, is ______.
Concept: undefined >> undefined
The molarity of the solution prepared by dissolving 6.3 g of oxalic acid (H2C2O4 · 2 H2O) in 250 mL of water in mol L-1 is x × 10-2. The value of x is ______. (Nearest integer)
[Atomic mass: H : 1.0, C : 12.0, O : 16.0]
Concept: undefined >> undefined
The mole fraction of a solute in a 100 molal aqueous solution is ______ × 10-2. (Round off to the Nearest Integer).
[Given :Atomic masses : H : 1.0 u, O : 16.0 u]
Concept: undefined >> undefined
A flask contains a mixture of compounds A and B. Both compounds decompose by first-order kinetics. The half-lives for A and B are 300 s and 180 s, respectively. If the concentrations of A and B are equal initially, the time required for the concentration of A to be four times that of B (in s) is ______. (Use ln 2 = 0.693)
Concept: undefined >> undefined
Match List - I with List - II.
| List - I | List - II | ||
| (One) | (Composition) | ||
| (A) | Siderite | (I) | FeCO3 |
| (B) | Malachite | (II) | CuCO3 · Cu(OH)2 |
| (C) | Sphalerite | (III) | ZnS |
| (D) | Calamine | (IV) | ZnCO3 |
Choose the correct answer from the options given below:
Concept: undefined >> undefined
Calamine and Malachite, respectively, are the ores of ______.
Concept: undefined >> undefined
The ore that contains both iron and copper is ______.
Concept: undefined >> undefined
4 ml of pure A (d = 2.45 gm/ml) was added to 46 ml of B (d = `25.1/23` gm/ml), the molarity of a solution of A in B will be ______, if the density of the final solution is 1.8 gm/ml.
Given: Molar mass of A = 98, Molar mass of B = 46
Concept: undefined >> undefined
The group number, number of valence electrons, and valency of an element with atomic number 15, respectively, are ______.
Concept: undefined >> undefined
The IUPAC symbol for the element with atomic number 119 would be ______.
Concept: undefined >> undefined
The one that will show optical activity is: (en = ethane 1, 2-diamine)
Concept: undefined >> undefined
ΔU is equal to ______.
Concept: undefined >> undefined
