Advertisements
Advertisements
For octahedral Mn(II) and tetrahedral Ni(II) complexes, consider the following statements:
- Both the complexes can be high spin.
- Ni(II) complex can very rarely below spin.
- With strong field Ligands, Mn(II) complexes can be low spin.
- Aqueous solution of Mn (II) ions is yellow in colour.
The correct statements are:
Concept: undefined >> undefined
The major product in the following reaction is:
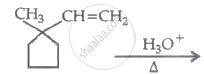
Concept: undefined >> undefined
Advertisements
Consider that d6 metal ion (M2+) forms a complex with aqua ligands and the spin only magnetic moment of the complex is 4.90 BM. The geometry and the crystal field stabilization energy of the complex are ______.
Concept: undefined >> undefined
The acceptable resonating structures of the following molecule are:
\[\begin{array}{cc}
\ce{CH3 - CH = C - \overset{\bullet\bullet}{\underset{\bullet\bullet}{O}} - CH2 - CH3}\\
|\phantom{......}\\
\ce{:N}\phantom{.......}\\
\phantom{}/\phantom{...}\backslash\phantom{......}\\
\phantom{}\ce{H3C}\phantom{.....}\ce{CH3}\phantom{......}
\end{array}\]
| (x) | \[\begin{array}{cc} \ce{CH3 - \overset{\overset{Θ}{\bullet\bullet}}{C}H - C - \overset{\bullet\bullet}{\underset{\bullet\bullet}{O}} - CH2 - CH3}\\ ||\phantom{......}\\ \ce{N^⊕}\phantom{.....}\\ \phantom{}/\phantom{...}\backslash\phantom{......}\\ \phantom{}\ce{H3C}\phantom{.....}\ce{CH3}\phantom{......} \end{array}\] |
| (y) | \[\begin{array}{cc} \ce{CH3 - \overset{\overset{Θ}{\bullet\bullet}}{C}H - C = \overset{⊕}{\underset{\bullet\bullet}{O}} - CH2 - CH3}\\ |\phantom{......}\\ \ce{:N}\phantom{.......}\\ \phantom{}/\phantom{...}\backslash\phantom{......}\\ \phantom{}\ce{H3C}\phantom{.....}\ce{CH3}\phantom{......} \end{array}\] |
| (z) | \[\begin{array}{cc} \ce{CH3 - \overset{⊕}{C}H - C - \overset{\bullet\bullet}{\underset{\bullet\bullet}{O}} - CH2 - CH3}\\ ||\phantom{......}\\ \ce{N^Θ}\phantom{.....}\\ \phantom{}/\phantom{...}\backslash\phantom{......}\\ \phantom{}\ce{H3C}\phantom{.....}\ce{CH3}\phantom{......} \end{array}\] |
| (w) | \[\begin{array}{cc} \ce{CH3 - \overset{⊕}{C}H - C = \overset{Θ}{O} - CH2 - CH3}\\ |\phantom{......}\\ \ce{:N}\phantom{.......}\\ \phantom{}/\phantom{...}\backslash\phantom{......}\\ \phantom{}\ce{H3C}\phantom{.....}\ce{CH3}\phantom{......} \end{array}\] |
Concept: undefined >> undefined
Isocyanide reaction involves the intermediate formation of ______.
Concept: undefined >> undefined
For the gaseous reaction, \[\ce{K_{(g)} + F_{(g)} -> K^+_{ (g)} + F^-_{ (g)}}\], ΔH was calculated to be 19 kcal/mol under conditions where the cations and anions were prevented by electrostatic separation from combining with each other. The ionisation energy of K is 4.3 eV. The electron affinity of F is ______. (in eV)
Concept: undefined >> undefined
The lassaigne's extract is boiled with dil. HNO3 before testing for halogens because ______.
Concept: undefined >> undefined
If the starting material is 1-methyl-1,2-epoxy cyclopentane, of absolute configuration, decide which one compound correctly represents the product of its reaction with sodium methoxide in methanol.
Concept: undefined >> undefined
`"A"_0/2` atoms of X(g) are converted into X+(g) by absorbing energy E1. `"A"_0/2` ions of X+(g) are converted into X−(g) with release of energy E2. Hence ionization energy and electron affinity of X(g) are ______.
Concept: undefined >> undefined
What happens when steam is passed over red-hot carbon?
Concept: undefined >> undefined
The decreasing order of the second ionization potential of K, Ca and Ba is ______.
Concept: undefined >> undefined
The presence of primary amines can be confirmed by ______.
Concept: undefined >> undefined
\[\ce{(A) ->[Cl2/hv] (B) ->[aq. KOH] (C) ->[O] CH3CHO}\]
Identify A, B and C:
Concept: undefined >> undefined
The vapour pressures of two volatile liquids A and B at 25°C are 50 Torr and 100 Torr, respectively. If the liquid mixture contains 0.3 mole fraction of A, then the mole fraction of liquid B in the vapour phase is `x/17`. The value of x is ______.
Concept: undefined >> undefined
Galvanization is applying a coating of ______.
Concept: undefined >> undefined
What are A and B in the following reaction?
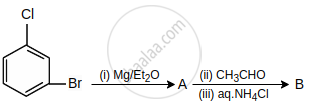
Concept: undefined >> undefined
Major product of the reaction is:
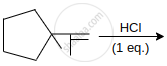
Concept: undefined >> undefined
Which of the following atoms has the highest first ionization energy?
Concept: undefined >> undefined
The complex that has highest crystal field splitting energy (Δ) is ______.
Concept: undefined >> undefined
Which of the following is not a correct statement for primary aliphatic amines?
Concept: undefined >> undefined
