Advertisements
Advertisements
The decomposition of formic acid on gold surface follows first-order kinetics. If the rate constant at 300 K is 1.0 × 10−3 s−1 and the activation energy Ea = 11.488 kJ mol−1, the rate constant at 200 K is ______ × 10−5 s−1. (Round off to the Nearest Integer)
(Given R = 8.314 J mol−1 K−1)
Concept: undefined >> undefined
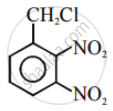
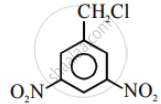
Among the following compounds I - IV, which one forms a yellow precipitate on reacting sequentially with (i) NaOH (ii) dil. HNO3 (iii) AgNO3?
 |
 |
 |
 |
| I | II | III | IV |
Concept: undefined >> undefined
Advertisements
Which of the following compounds will show retention in configuration on nucleophile substitution by OH− ion?
Concept: undefined >> undefined
Maltose on treatment with dilute HC?
Concept: undefined >> undefined
The number of chiral carbons present in the molecule given below is ______.

Concept: undefined >> undefined
\[\ce{X + C + Cl2 ->[High temperature][of about 1000 K] Y + CO}\];
\[\ce{Y + 2H2O -> Z + 2HCl}\]
Compound Y is found in polymeric chain structure and is an electron-deficient Molecule. Y must be:
Concept: undefined >> undefined
Gold is extracted by making soluble cyanide complex. The cyanide complex is ______.
Concept: undefined >> undefined
A definite volume of H2O2 undergoing spontaneous decomposition required 22.8 c.c. of standard permanganate solution for titration. After 10 and 20 minutes respectively the volumes of permanganate required were 13.8 and 8.25 c.c. The time required for the decomposition to be half completed is ______ min.
Concept: undefined >> undefined
The reaction \[\ce{SO2Cl2(g) -> SO2(g) + Cl2(g)}\] is a first-order gas reaction with k = 2.2 × 10−5 sec−1 at 320°C. The percentage of SO2Cl2 is decomposed on heating this gas for 90 min, is ______%.
Concept: undefined >> undefined
For a first order reaction, the ratio of the time for 75% completion of a reaction to the time for 50% completion is ______. (Integer answer)
Concept: undefined >> undefined
Which of the reaction is suitable for concentrating ore by leaching process?
Concept: undefined >> undefined
In SN1 reactions, the correct order of reactivity for the following compounds:
CH3Cl, CH3CH2Cl, (CH3)2CHCl and (CH3)3CCl is ______.
Concept: undefined >> undefined
If a rocket runs on fuel (C15 H30) and liquid oxygen, the weight of oxygen required and CO2 released for every Iitre of fuel respectively are ______.
(Given: density of the fuel is 0.756 g/mL)
Concept: undefined >> undefined
What volume of hydrogen gas, at 273 K and 1 atm pressure will be consumed in obtaining 21.6 g of elemental boron (atomic mass = 10.8) from the reduction of boron trichloride by hydrogen?
Concept: undefined >> undefined
The bond order in NO is 2.5 while that in NO+ is 3. Which of the following statements is true for these two species?
Concept: undefined >> undefined
Bond distance in HF is 9.17 × 10−11 m. Dipole moment of HF is 6.104 × 10−30 Cm. The percentage of ionic character in HF will be ______.
(electron charge = 1.60 × 10−19 C)
Concept: undefined >> undefined
Optical activity of an enantiomeric mixture is +12.6° and the specific rotation of (+) isomer is +30°. The optical purity is ______ %.
Concept: undefined >> undefined
The decreasing order of reactivity of the following compounds towards nucleophilic substitution (SN2) is ______.
Concept: undefined >> undefined
A solution of phenol in chloroform when treated with aqueous NaOH gives compound P as a major product. The mass percentage of carbon in P is ______. (to the nearest integer) (Atomic mass: C = 12; H = 1; O = 16)
Concept: undefined >> undefined
Which of the following will not show mutarotation?
Concept: undefined >> undefined