Advertisements
Advertisements
The mass of gas adsorbed, x per unit mass of adsorbed, m was measured at various x pressures p. A graph between `log "x"/"m"` and log p gives a straight line with slope equal to 2 and the intercept equal to 0.4771. The value of `"x"/"m"` at a pressure of 4 atm is ______.
[Given log 3 = 0.4771]
Concept: undefined >> undefined
Adsorption of a gas follows Freundlich adsorption isotherm. x is the mass of the gas adsorbed on mass m of the adsorbent. The x plot of log `x/m` versus log p is shown in the given graph. `x/m` is proportional to ______.
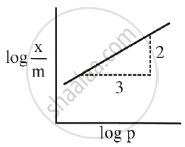
Concept: undefined >> undefined
Advertisements
For silver Cp (J K-1 mol-1) = 23 + 0.01 T. If the temperature (T) of 3 moles of silver is raised from 300 K to 1000 K at 1 atom pressure, the value of ΔH will be close to ______.
Concept: undefined >> undefined
\[\ce{Y <-[\Delta, 205°C] CaSO4.2H2O ->[\Delta, 120°C] X}\]
X and Y are respectively:
Concept: undefined >> undefined
Alkylamine dissolve in hydrochloric acid to form alkylammonium chloride. The nitrogen in the latter salt is ______.
Concept: undefined >> undefined
A 0.5 g sample of an iron-containing mineral mainly in the form of CuFeS2 was reduced suitably to convert all the ferric iron into ferrous form and was obtained as a solution. In the absence of any interfering matter, the solution required 42 ml of 0.01 M K2Cr2O7 solution for titration. The percentage of CuFeS2 in the mineral is ______%.
(Cu = 63.5, Fe = 55.8, S = 32, O = 16)
Concept: undefined >> undefined
Calculate the work involved when 1 mol of an ideal gas is compressed reversibly from 1.00 bar to 5.00 bar at a constant temperature of 300 K ______.
Concept: undefined >> undefined
What would be the main product when propene reacts with HBr in presence of benzoyl peroxide?
Concept: undefined >> undefined
The net work done in the following cycle for one mol of an ideal gas will be ______ (in calorie), where in process BC, PT = constant. (R = 2 cal/mol-K).

Concept: undefined >> undefined
1 mole of an ideal monoatomic gas initially at 1 atm and 300 K experiences a process by which pressure is doubled. The nature of the process is unspecified but 6. ΔU = 900 cal. The final volume will be ______ l.
[Given : R = 0.08 atm lit. I mol/K = 2 Cal/K/mol J]
Concept: undefined >> undefined
1 mole of an ideal monoatomic gas initially at 1 atm and 300 K experiences a process by which pressure is doubled. The nature of the process is unspecified but 6. ΔU = 900 cal. The final volume will be ______ l.
[Given : R = 0.08 atm lit. I mol/K = 2 Cal/K/mol J]
Concept: undefined >> undefined
For Freundlich adsorption isotherm, a plot of log (x/m) (Y-axis) and log p (x-axis) gives a straight line. The intercept and slope for the line is 0.4771 and 2, respectively. The mass of gas, adsorbed per gram of adsorbent if the initial pressure is 0.04 atm, is ______ × 10-4 g. (log 3 = 0.4771)
Concept: undefined >> undefined
Find the work done when 2 moles of hydrogen expand isothermally from 15 to 50 litres against a constant pressure of 1 atm at 25°C.
Concept: undefined >> undefined
Arrange the following in increasing order of their covalent character.
(A) CaF2
(B) CaCl2
(C) CaBr2
(D) CaI2
Choose the correct answer from the options given below.
Concept: undefined >> undefined
Lattice energy of an ionic compound depends upon ______.
Concept: undefined >> undefined
A compound 'X' is acidic and it is soluble in NaOH solution, but insoluble in NaHC0 3 solution. Compound 'X' also gives violet colour with neutral FeCI3 solution. The compound 'X' is:
Concept: undefined >> undefined
The interaction energy of London forces between two particles is proportional to rx, where r is the distance between the particles. The value of x is ______.
Concept: undefined >> undefined
Match List - I with List - II.
| List I | List II | ||
| (A) |  |
(I) | Br2 in CS2 |
| (B) | (II) | Na2Cr2O7/H2SO4 | |
| (C) |  |
(III) | Zn |
| (D) |  |
(IV) | CHCl3/NaOH |
Choose the correct answer from the options given below:
Concept: undefined >> undefined
The predominant intermolecular forces present in ethyl acetate, a liquid, are ______.
Concept: undefined >> undefined
Five moles of an ideal gas at 1 bar and 298 K is expanded into the vacuum to double the volume. The work done is ______.
Concept: undefined >> undefined
