Advertisements
Advertisements
Which one of the following equations does not correctly represent the first law of thermodynamics for the given processes involving an ideal gas? (Assume non-expansion work is zero)
Concept: undefined >> undefined
Which of the following amines can be prepared by Gabriel phthalimide reaction?
Concept: undefined >> undefined
Advertisements
Which of the following compound is optically active?
Concept: undefined >> undefined
An inorganic salt solution gives a yellow precipitate with silver nitrate. The precipitate dissolves in dilute nitric acid as well as in ammonium hydroxide. The solution contains _______.
Concept: undefined >> undefined
The major product of the following reaction is:
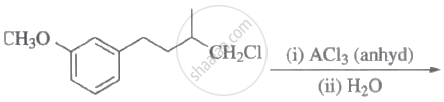
Concept: undefined >> undefined
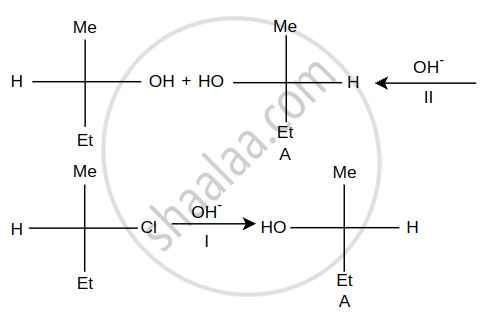
Steps I and II are ______.
Concept: undefined >> undefined
Which of the following statements is correct for CsBr3?
Concept: undefined >> undefined
In Finkelstein Reaction, which reactants are used?
Concept: undefined >> undefined
\[\ce{BaC2 + N2 ->[\Delta] (1)}\]
\[\ce{CaC2 + N2 ->[\Delta] (2)}\] (1) and (2) are ______.
Concept: undefined >> undefined
Consider the following carbocations:


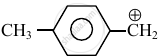
- \[\ce{CH3 - \overset{⊕}{CH2}}\]
The relative stabilities of these carbocations are such that:
Concept: undefined >> undefined
\[\ce{C2H5Cl + AgF -> C2H5F + AgCl}\] The above reaction is called ______.
Concept: undefined >> undefined
Ca, Ba and Sr ions are precipitated in fifth group as their ______.
Concept: undefined >> undefined
Which compound would exhibit optical isomers?
Concept: undefined >> undefined
The number of geometrical isomers of \[\ce{[Co(NH3)3 (NO3)3]}\] are ______.
Concept: undefined >> undefined
For a reaction of order n, the unit of the rate constant is ______.
Concept: undefined >> undefined
Halides of alkaline earth metals form hydrate such as MgCl2.6H2O, CaCl2.6H2O, BaCl2.2H2O and SrCl2.2H2O. This shows that halides of group 2 elements:
Concept: undefined >> undefined
When sucrose is heated with cone. HNO3 the product is ______.
Concept: undefined >> undefined
The major product of the following reaction is:
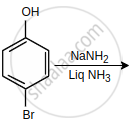
Concept: undefined >> undefined
White precipitate of AgCl dissolves in aqueous ammonia solution due to formation of ______.
Concept: undefined >> undefined
The one that is not expected to show isomerism is ______.
Concept: undefined >> undefined
