Advertisements
Advertisements
Affinity for hydrogen decreases in the group from fluorine to iodine. Which of the halogen acids should have the highest bond dissociation enthalpy?
Concept: undefined >> undefined
On heating compound (A) gives a gas (B) which is a constituent of air. This gas when treated with 3 mol of hydrogen (H2) in the presence of a catalyst gives another gas (C) which is basic in nature. Gas C on further oxidation in moist condition gives a compound (D) which is a part of acid rain. Identify compounds (A) to (D) and also give necessary equations of all the steps involved.
Concept: undefined >> undefined
Advertisements
Read the passage carefully and answer the questions that follow:
|
Strengthening the Foundation: Chargaff Formulates His "Rules" Many people believe that James Watson and Francis Crick discovered DNA in the 1950s. In reality, this is not the case. Rather, DNA was first identified in the late 1860s by Swiss chemist Friedrich Miescher. Then, in the decades following Miescher's discovery, other scientists- notably, Phoebus Levene and Erwin Chargaff- carried out a series of research efforts that revealed additional details about the DNA molecule, including its primary chemical components and the ways in which they joined with one another. Without the scientific foundation provided by these pioneers, Watson and Crick may never have reached their groundbreaking conclusion of 1953: that the DNA molecule exists in the form of a three-dimensional double helix. |
Answer the following questions:
- A segment of DNA has 100 adenine and 150 cytosine bases. What is the total number of nucleotides present in this segment of DNA?
- A sample of hair and blood was found at two sites. Scientists claim that the samples belong to same species. How did the scientists arrive at this conclusion?
- The sample of a virus was tested and it was found to contain 20% adenine, 20% thymine, 20% guanine and the rest cytosine. Is the genetic material of this virus (a) DNA-double helix (b) DNA-single helix (c) RNA? What do you infer from this data?
OR
How can Chargaff’s rule be used to infer that the genetic material of an organism is double-helix or single-helix?
Concept: undefined >> undefined
Write the main product in the following reaction:
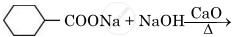
Concept: undefined >> undefined
DNA fingerprinting is used to determine paternity of an individual. Which property of DNA helps in the procedure?
Concept: undefined >> undefined
Account for the following:
Fluorine forms only one oxoacid HOF.
Concept: undefined >> undefined
Draw the structures of the following:
(1) HClO4
(2) H3PO3
Concept: undefined >> undefined
When RNA is hydrolysed, there is no relationship among the quantities of different bases obtained. What does this fact suggest about the structure of RNA?
Concept: undefined >> undefined
Draw the structures of the HClO3.
Concept: undefined >> undefined
Account for the following :
HClO4 is a stronger acid than HClO.
Concept: undefined >> undefined
The increasing order of reducing power of the halogen acids is:
Concept: undefined >> undefined
Which of the following statements is true for protein synthesis?
Concept: undefined >> undefined
Which one of the following order is correct for the bond dissociation enthalpy of halogen molecule?
Concept: undefined >> undefined
Assertion: HI cannot be prepared by the reaction of KI with concentrated H2SO4.
Reason: HI has the lowest H-X bond strength among halogen acids.
Concept: undefined >> undefined
A black compound of manganese reacts with a halogen acid to give greenish-yellow gas. When excess of this gas reacts with \[\ce{NH3}\] an unstable trihalide is formed. In this process the oxidation state of nitrogen changes from ______.
Concept: undefined >> undefined
Explain why the stability of oxoacids of chlorine increases in the order given below:
\[\ce{HClO < HClO2 < HClO3 < HClO4}\]
Concept: undefined >> undefined
Assertion: \[\ce{HI}\] cannot be prepared by the reaction of KI with concentrated \[\ce{H2SO4}\]
Reason: \[\ce{HI}\] has lowest \[\ce{H - X}\] bond strength among halogen acids.
Concept: undefined >> undefined
Draw the structures of the following molecules: BrF3
Concept: undefined >> undefined
ICl is more reactive than I2.
Concept: undefined >> undefined
Account for the following: Interhalogens are more reactive than pure halogens.
Concept: undefined >> undefined
