Advertisements
Advertisements
Preparation of alcohols from alkenes involves the electrophilic attack on alkene carbon atom. Explain its mechanism.
Concept: undefined >> undefined
Explain why low molecular mass alcohols are soluble in water.
Concept: undefined >> undefined
Advertisements
\[\begin{array}{cc}
\ce{CH3 - C ≡ CH ->[40{%} H2SO4][1{%} HgSO4] A ->[Isomerisation] CH3 - C - CH3}\\
\phantom{........................................}||\\
\phantom{........................................}\ce{O}\\
\end{array}\]
Structure of ‘A’ and type of isomerism in the above reaction are respectively.
Concept: undefined >> undefined
Amino acids are classified as acidic, basic or neutral depending upon the relative number of amino and carboxyl groups in their molecule. Which of the following are acidic?
(i)
\[\begin{array}{cc}
\ce{(CH3)2CH - CH - COOH}\\
\phantom{..}|\\
\phantom{.....}\ce{NH2}
\end{array}\]
(ii)
\[\begin{array}{cc}
\ce{HOOC - CH2 - CH2 - CH - COOH}\\
\phantom{............}|\\
\phantom{...............}\ce{NH2}
\end{array}\]
(iii)
\[\ce{H2N - CH2 - CH2 - CH2 - COOH}\]
(iv)
\[\begin{array}{cc}
\ce{HOOC - CH2 - CH - COOH}\\
\phantom{.....}|\\
\phantom{........}\ce{NH2}
\end{array}\]
Concept: undefined >> undefined
Assertion: Glycine must be taken through diet.
Reason: It is an essential amino acid.
Concept: undefined >> undefined
Which of the following polymers have vinylic monomer units?
(i) Acrilan
(ii) Polystyrene
(iii) Nylon
(iv) Teflon
Concept: undefined >> undefined
How is the following resin intermediate prepared and which polymer is formed by this monomer unit?
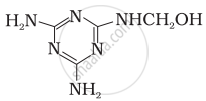
(Resin intermediate)
Concept: undefined >> undefined
Match the polymers given in Column I with their commercial names given in Column II.
| Column I | Column II |
| (i) Polyester of glycol and phthalic acid | (a) Novolac |
| (ii) Copolymer of 1, 3-butadiene and styrene | (b) Glyptal |
| (iii) Phenol and formaldehyde resin | (c) Buna-S |
| (iv) Polyester of glycol and terephthalic acid | (d) Buna-N |
| (v) Copolymer of 1, 3-butadiene and acrylonitrile | (e) Dacron |
Concept: undefined >> undefined
Which of the following polymers soften on heating and harden on cooling? What are the polymers with this property collectively called? What are the structural similarities between such polymers? Bakelite, urea-formaldehyde resin, polythene, polyvinyls, polystyrene.
Concept: undefined >> undefined
How would you account for the following:
There is a greater range of oxidation states among the actinoids than among the lanthanides.
Concept: undefined >> undefined
Name the suitable alcohol and reagent, from which 2-Chloro-2-methyl propane can be prepared.
Concept: undefined >> undefined
Write the equations for the following reaction:
Tert butyl chloride is treated with sodium ethoxide.
Concept: undefined >> undefined
Why does the cell voltage of a mercury cell remain constant during its lifetime?
Concept: undefined >> undefined
Write any two consequences of Lanthanoid Contraction.
Concept: undefined >> undefined
Which of the following reactions are feasible?
Concept: undefined >> undefined
Give reasons:
Mercury cell delivers a constant potential during its life time.
Concept: undefined >> undefined
Write the mechanism of the following reaction:
\[\ce{2CH3CH2OH ->[H^+][413 K] CH3-CH2-O-CH2-CH3 + H2O}\]
Concept: undefined >> undefined
Draw the zwitter ion structure for sulphanilic acid.
Concept: undefined >> undefined
Give a reason for the following observations:
Amino acids behave like salts.
Concept: undefined >> undefined
A non-volatile solute 'X' (molar mass = 50 g mol−1), when dissolved in 78 g of benzene, reduced its vapour pressure to 90%. Calculate the Mass of X dissolved in the solution.
Concept: undefined >> undefined
