Please select a subject first
Advertisements
Advertisements
The following compound can be called as:

Concept: undefined >> undefined
The spatial arrangement of the given molecule is denoted by:

Concept: undefined >> undefined
Advertisements
Choose the appropriate answer(s) for the below representation from the options given

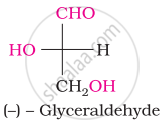
Concept: undefined >> undefined
The number of asymmetric carbon atom(s) below the figure is/are

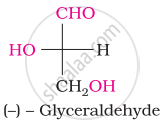
Concept: undefined >> undefined
What is the most abundant organic compound on earth?
Concept: undefined >> undefined
Oxime is formed by treating glucose with ____________.
Concept: undefined >> undefined
Acetylation of glucose yields ____________.
Concept: undefined >> undefined
Glucose does not give Schiff’s test because of the formation of cyclic ____________.
Concept: undefined >> undefined
The space lattice of graphite is ____________.
Concept: undefined >> undefined
Coordination numbers of Zn2+ and S2– in the crystal structure of wurtzite are ____________.
Concept: undefined >> undefined
In face-centered cubic lattice, a unit cell is shared equally by how many unit cells?
Concept: undefined >> undefined
The total number of tetrahedral voids in the face-centered unit cell is ______.
Concept: undefined >> undefined
Which of the following factor do not affect the solubility of a solid solute in liquid?
Concept: undefined >> undefined
When a solid solute is added to the solvent, some solute dissolves and its concentration increases in solution. This process is known as ___________. Some solute particles in the solution collide with the solid solute particles and get separated out of the solution. This process is known as __________.
Concept: undefined >> undefined
At the state of dynamic equilibrium, for solute + solvent solution.
Concept: undefined >> undefined
Which of the following statements is incorrect?
Concept: undefined >> undefined
On dissolving sugar in water at room temperature solution feels cool to touch. Under which of the following cases dissolution of sugar will be most rapid?
Concept: undefined >> undefined
A beaker contains a solution of the substance ‘A’. Precipitation of substance ‘A’ takes place when a small amount of ‘A’ is added to the solution. The solution is ______.
Concept: undefined >> undefined
Maximum amount of a solid solute that can be dissolved in a specified amount of a given liquid solvent does not depend upon ______.
Concept: undefined >> undefined
The catalyst used in the manufacture of HNO3 by Ostwald’s process is ____________.
Concept: undefined >> undefined
