Advertisements
Advertisements
Rohan heard that instructions were given to the laboratory attendent to store a particular chemical i.e., keep it in the darkroom, add some urea in it, and keep it away from dust. This chemical acts as an oxidising as well as a reducing agent in both acidic and alkaline media. This chemical is important for use in the pollution control treatment of domestic and industrial effluents. Write the name of this compound.
Concept: undefined >> undefined
Rohan heard that instructions were given to the laboratory attendent to store a particular chemical i.e., keep it in the darkroom, add some urea in it, and keep it away from dust. This chemical acts as an oxidising as well as a reducing agent in both acidic and alkaline media. This chemical is important for use in the pollution control treatment of domestic and industrial effluents. Write the name of this compound. Explain why such precautions are taken for storing this chemical.
Concept: undefined >> undefined
Advertisements
What is the importance of heavy water?
Concept: undefined >> undefined
Give a method for the manufacture of hydrogen peroxide and explain the reactions involved therein.
Concept: undefined >> undefined
What mass of hydrogen peroxide will be present in 2 litres of a 5 molar solution? Calculate the mass of oxygen which will be liberated by the decomposition of 200 mL of this solution.
Concept: undefined >> undefined
Silicon has a strong tendency to form polymers like silicones. The chain length of silicone polymer can be controlled by adding ______.
Concept: undefined >> undefined
Me3SiCl is used during polymerisation of organo silicones because:
(i) the chain length of organo silicone polymers can be controlled by adding Me3SiCl.
(ii) Me3SiCl blocks the end terminal of silicone polymer.
(iii) Me3SiCl improves the quality and yield of the polymer.
(iv) Me3SiCl acts as a catalyst during polymerisation.
Concept: undefined >> undefined
Assertion (A): Silicons are water repelling in nature.
Reason (R): Silicons are organosilicon polymers, which have (–R2SiO–) as repeating unit.
Concept: undefined >> undefined
What are silicones? State the uses of silicones.
Concept: undefined >> undefined
In which of the following representations given below spatial arrangement of group/ atom different from that given in structure ‘A’?
 |
| (A) |
| (i) | 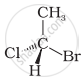 |
| (ii) |  |
| (iii) | 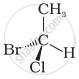 |
| (iv) | 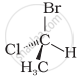 |
Concept: undefined >> undefined
What is the hybridisation of each carbon in H2C = C = CH2.
Concept: undefined >> undefined
Show the polarisation of carbon-magnesium bond in the following structure.
CH3 – CH2 – CH2 – CH2 – Mg – X
Concept: undefined >> undefined
Draw the possible resonance structures for \[\ce{CH3 - \overset{\bullet\bullet}{\underset{\bullet\bullet}{O}} - \overset{+}{C}H2}\] and predict which of the structures is more stable. Give reason for your answer.
Concept: undefined >> undefined
Which of the following ions is more stable? Use resonance to explain your answer.

Concept: undefined >> undefined
The structure of triphenylmethyl cation is given below. This is very stable and some of its salts can be stored for months. Explain the cause of high stability of this cation.

Concept: undefined >> undefined
Draw the resonance structure of the following compounds;
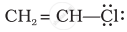
Concept: undefined >> undefined
Draw the resonance structure of the following compounds;
CH2 = CH – CH = CH2
Concept: undefined >> undefined
Draw the resonance structure of the following compounds;
\[\begin{array}{cc}
\ce{CH2 = CH - C = O}\\
\phantom{.........}|\\
\phantom{.........}\ce{H}
\end{array}\]
Concept: undefined >> undefined
Which of the following compounds will not exist as resonance hybrid. Give reason for your answer:
Concept: undefined >> undefined
Resonance structures of propenal are given below. Which of these resonating structures is more stable? Give reason for your answer.
\[\ce{\underset{I}{CH2 = CH - CH = O} <-> \underset{II}{\overset{⊕}{C}H2 - CH = CH - \overset{Θ}{O}}}\]
Concept: undefined >> undefined
