Definitions [5]
The branch of chemistry that deals with compounds originally derived from living organisms, such as sugar, starch, proteins, and acetic acid.
or
Organic compounds are the hydrides of carbon (hydrocarbons) and their derivatives. The branch of chemistry dealing with these compounds is called organic chemistry.
The branch of chemistry that deals with compounds obtained from non-living sources or minerals, such as common salt, blue vitriol, and nitrates.
Nomenclature is the system of assignment of names to organic compounds.
Compounds having the same molecular formula but different structural formula are known as Isomers and the phenomenon is known as Isomerism. (iso = same, meros = parts).
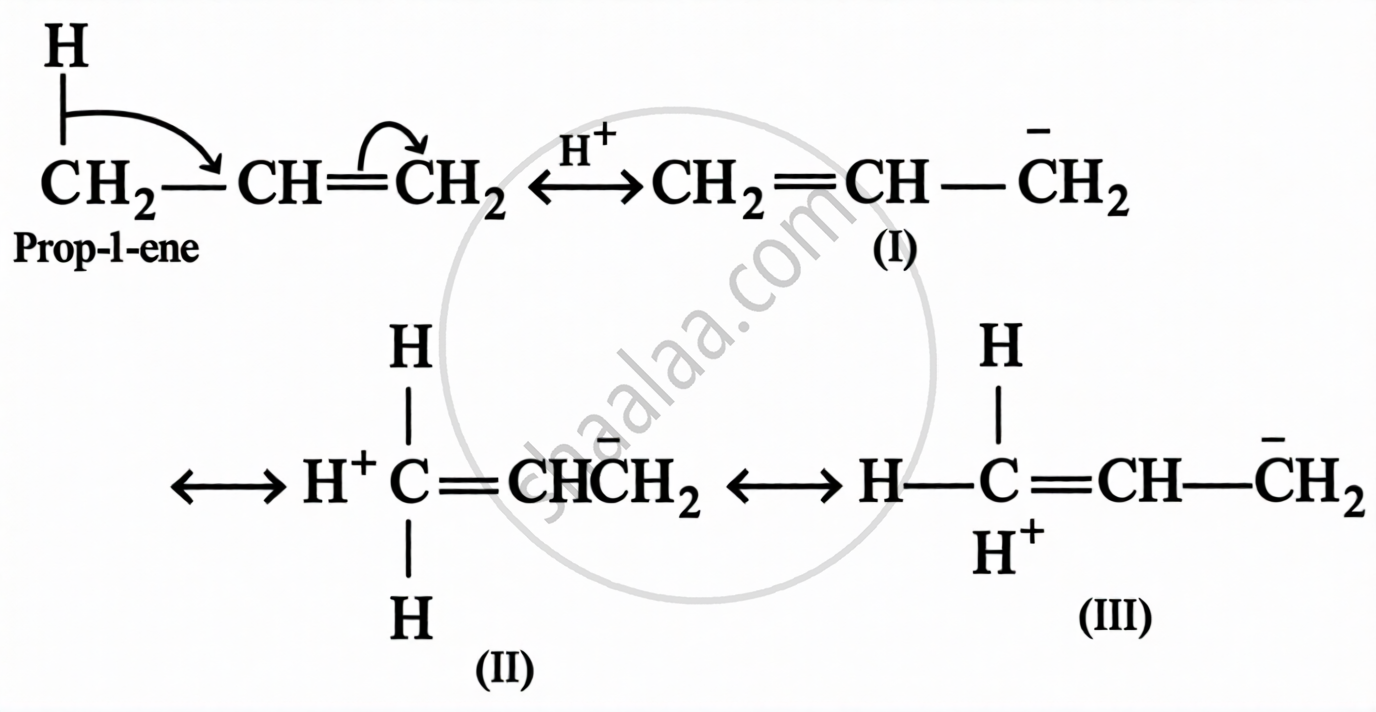
The stabilising interaction that involves delocalisation of σ-electrons of C–H bond of an alkyl group linked directly to an atom of unsaturated system or to an atom having unshared p-orbital, is called hyperconjugation.
e.g.
Key Points
2D Representations
| Type | Description | Example |
|---|---|---|
| Dash (Structural) Formula | Shows all atoms and all covalent bonds by dashes |
\[\begin{array}{cc} |
| Condensed Formula | Groups atoms together without showing all bonds | CH₃CH₂CH₃ or CH₃–CH₂–CH₃ |
| Bond Line / Zig-Zag Formula | Skeletal structure — lines represent C–C bonds; carbon atoms at line intersections and ends; H atoms implied |
3D Representations
| Type | Description |
|---|---|
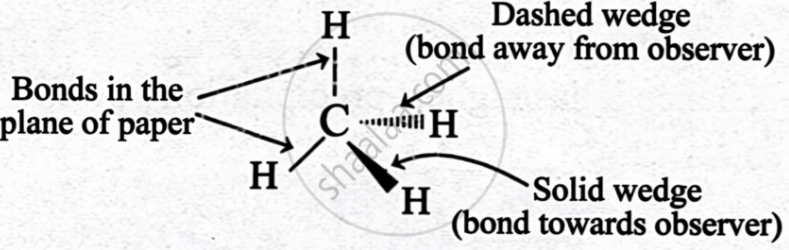
| Wedge Formula |
Solid wedge
|
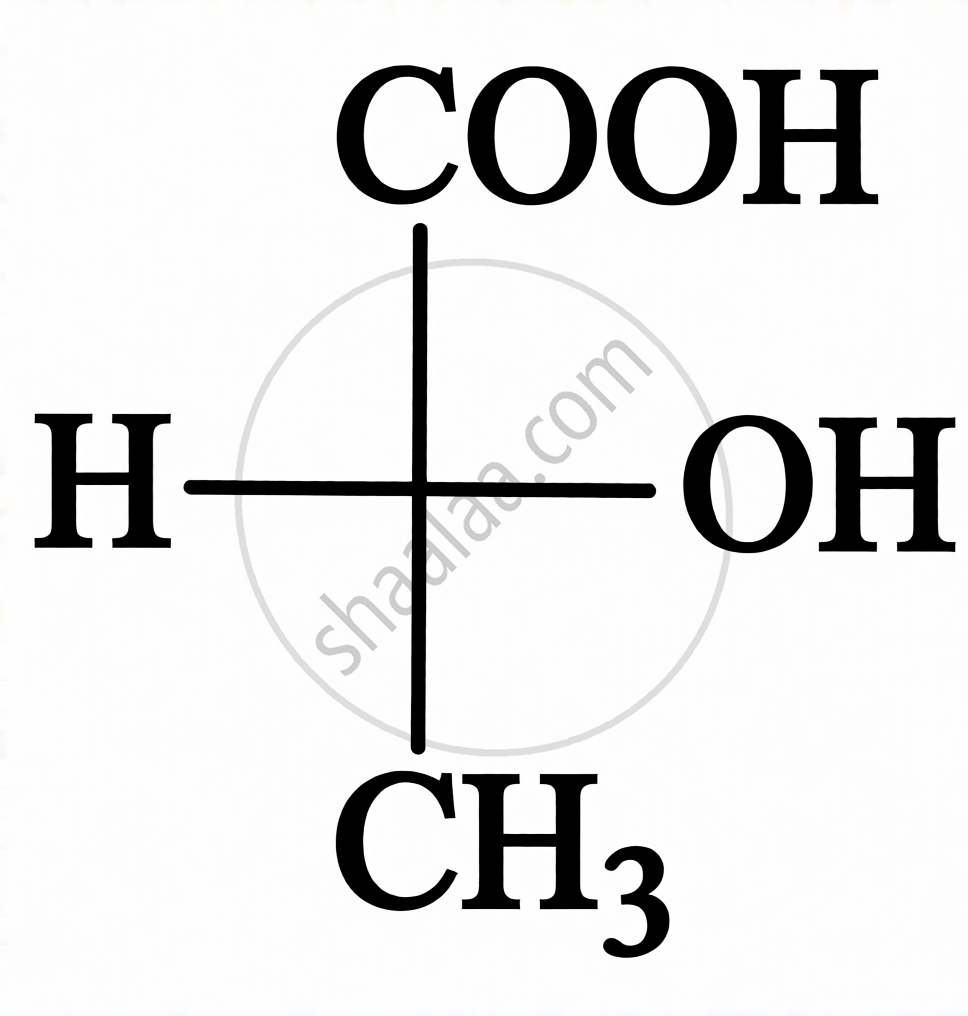
| Fischer Projection (Cross Formula) |
Carbon chain drawn vertically; horizontal lines represent bonds projecting up, vertical lines represent bonds going below the carbon.
|
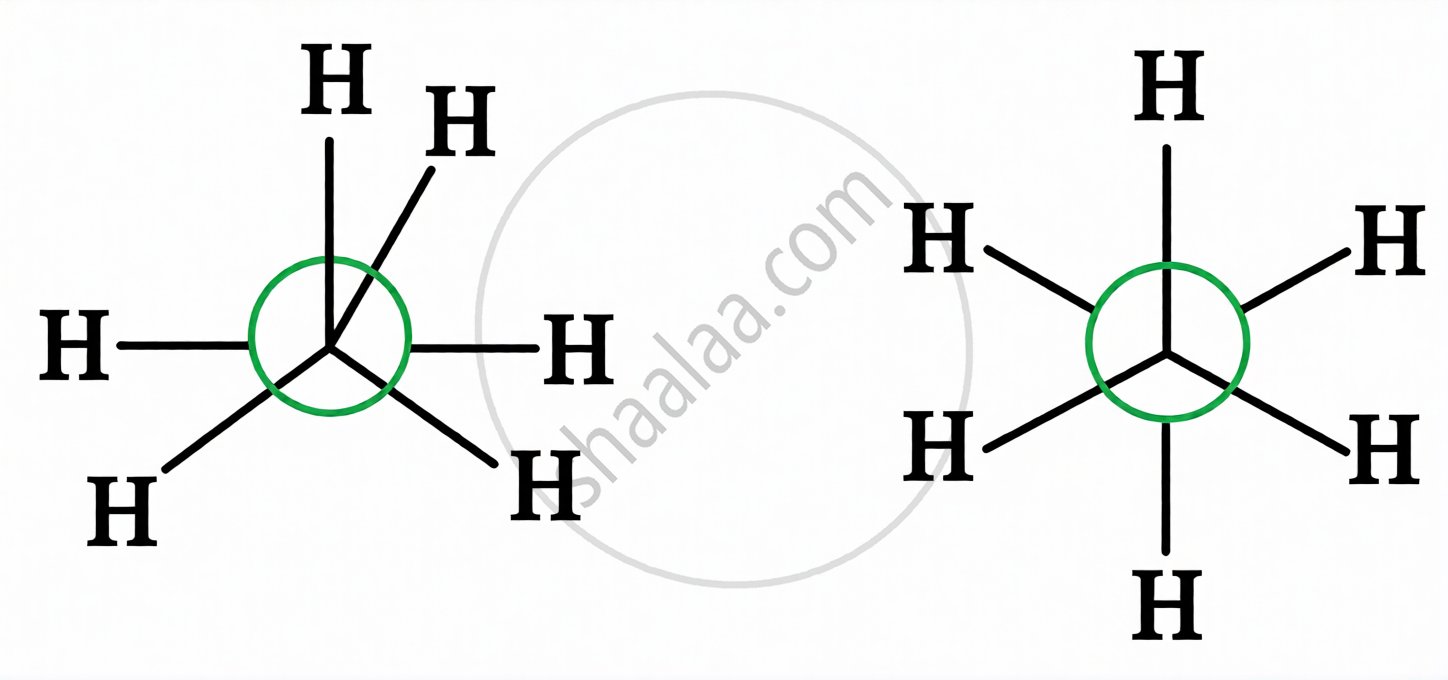
| Newman Projection |
Front carbon shown as a point, rear carbon as a circle; remaining 3 bonds at each carbon drawn as spokes of a wheel.
|
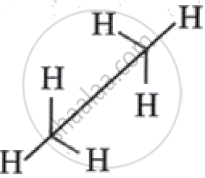
| Sawhorse / Perspective Formula |
Three-dimensional representation showing spatial arrangement.
|
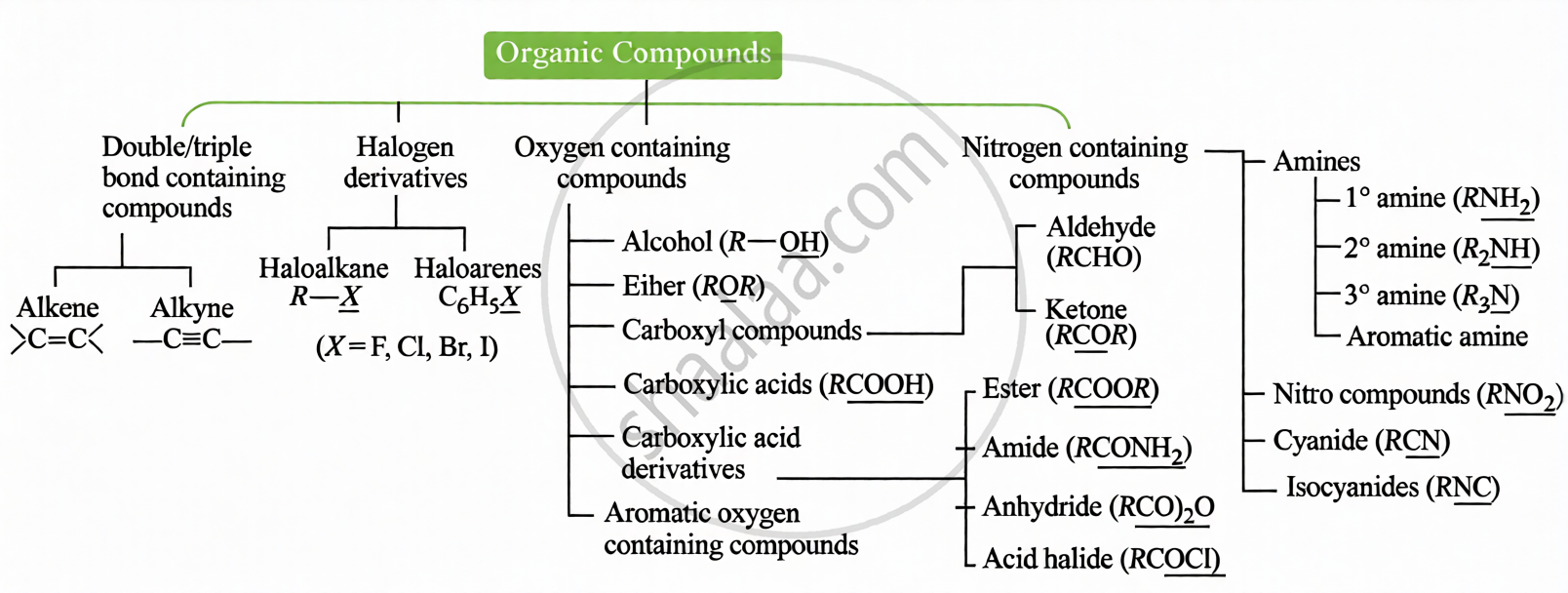
(i) Based on Functional Group:
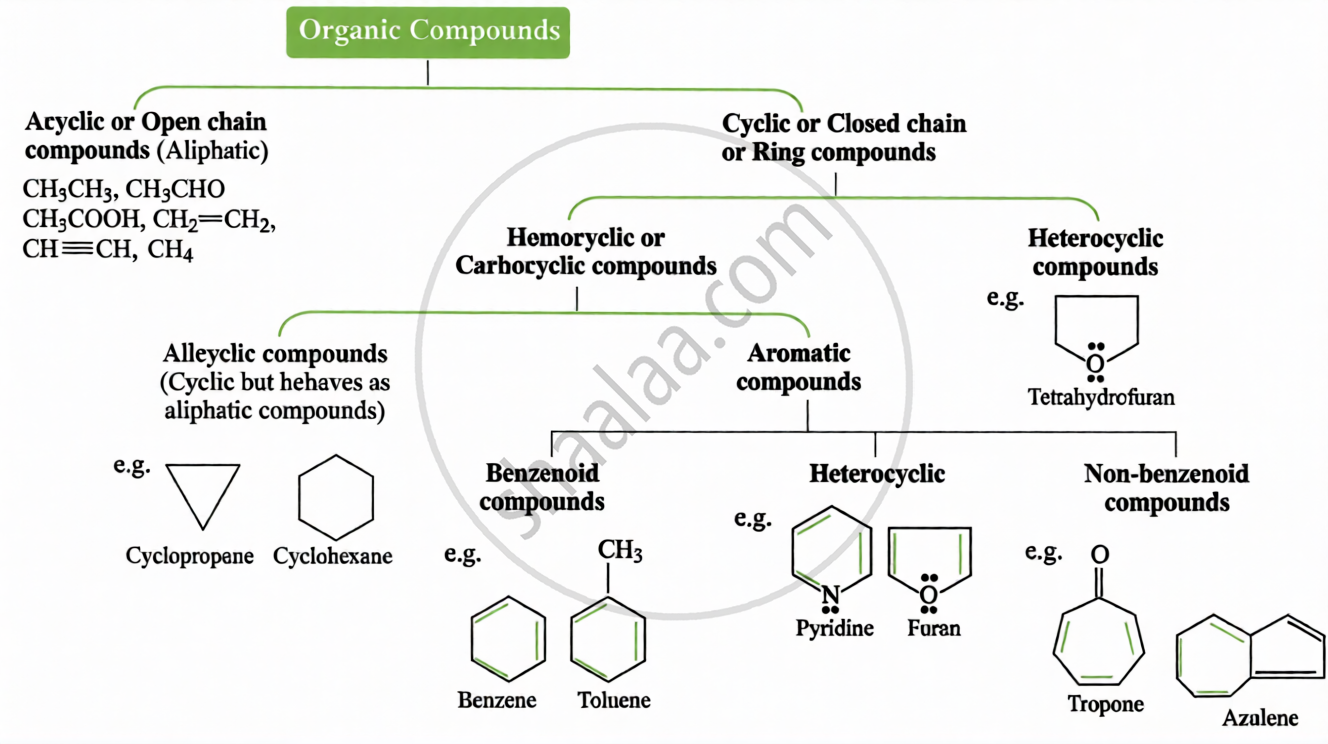
(ii) Based on Carbon Skeleton:
- The IUPAC system provides a unique, systematic way to name carbon compounds based on structure, replacing confusing common names.
- An IUPAC name has three parts: prefix, parent, and suffix, reflecting the carbon chain and functional group.
- The parent name is based on the longest carbon chain, and its ending changes to –ane, –ene, or –yne depending on the number of bonds.
- Functional groups are shown as prefixes or suffixes, and the chain is numbered to give them the lowest possible number.
- If the suffix begins with a vowel, the final ‘e’ in the parent alkane name is dropped (e.g., propane → propanone).
- Longest chain rule — The longest continuous C–C linkage is the parent chain
- Lowest locant rule — Number the parent chain from the end that gives substituents the lowest possible locant numbers
- Alphabetical order rule — When multiple substituents are present, name them in alphabetical order (ignoring multiplying prefixes like di, tri)
- First point of difference rule — If functional groups are at equivalent positions, number so that the first different locant is the lowest
- Complex group rule — For branched side chains, the carbon through which the complex group is attached to the main chain is always given locant number 1
- Double and triple bond priority — If both are at equal distance from ends, double bond is preferred; if asymmetrical, use lowest locant sum rule then first point of difference rule
- Multiple functional groups — Priority order: –COOH > –SO₃H > –COOR > –COCl > –CONH₂ > –CN > –CHO > C=O > –OH > –NH₂ > C=C > –C≡C–
1. Structural Isomerism
Same molecular formula but different connectivity (bonding) of atoms.
Types:
- Chain isomerism: Different carbon skeleton (straight/branched)
- Position isomerism: Functional group at different positions
- Functional isomerism: Different functional groups (e.g., alcohol vs ether)
- Metamerism: Different alkyl groups on either side of same functional group
- Tautomerism: Dynamic equilibrium between two forms (keto ↔ enol)
2. Stereoisomerism
Same molecular formula and bonding but different spatial arrangement.
Types:
I. Geometrical isomerism:
- Due to restricted rotation (double bond)
- Forms cis (same side) and trans (opposite side)
II. Optical isomerism:
- Mirror image isomers (enantiomers)
- Show optical activity (rotate plane polarized light)
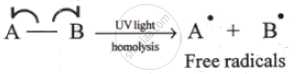
| Homolytic cleavage | Heterolytic cleavage |
|---|---|
| The symmetrical breaking of a covalent bond between two atoms such that each atom retains one electron of the shared pair. | The unsymmetrical breaking of a covalent bond between two atoms in such a way that the more electronegative atom acquires both the electrons of the shared pair. |
| Formation of free radicals (uncharged species) bearing unpaired electrons takes place. | Formation of charged species called ions, like carbocation or carbanion takes place. |
 |
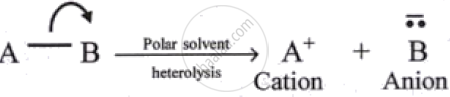 |
| Favourable in a non-polar solvent. | Favourable in a polar solvent. |
1. Electrophiles (E⁺) — Electron-loving
Attack regions of high electron density.
| Type | Examples |
|---|---|
| Neutral electrophiles | Free radicals (R- ), SO₃, BF₃, AlCl₃, NR (nitrenes), CR₂ (carbenes) |
| Positively charged electrophiles | H⁺, Cl⁺ (chloronium ion), I⁺, Br⁺, NO⁺ (nitrosonium ion), R⁺ (carbocation), NO₂⁺ |
2. Nucleophiles (Nu⁻) — Nucleus-loving
Attack regions of low electron density (electron-deficient centres).
| Type | Examples |
|---|---|
| Neutral nucleophiles | H₂O:, R₃N, R₂NH, NH₃, etc. |
| Negatively charged nucleophiles | Br⁻, CN⁻, NO₂⁻, C₆H₅O⁻, RCOO⁻, OR⁻, etc. |
- Permanent effect due to shift of σ-electrons
- Electrons move towards more electronegative atom
- Effect decreases with distance (negligible after 3 carbons)
e.g-
![]()
| Type | Meaning | Example |
|---|---|---|
| –I effect | Electron withdrawing group | –NO₂, –Cl |
| +I effect | Electron donating group | –CH₃ |
- Resonance occurs when a single Lewis structure cannot accurately describe a molecule.
- Multiple structures (canonical structures) differing only in the arrangement of electrons (not atoms) are drawn.
- The actual molecule is a resonance hybrid — a weighted average of all canonical structures.
- Resonance energy = Actual bond energy − Energy of most stable resonating structure.
Conditions for Writing Resonance Structures:
- Same atomic positions in all structures.
- Same number of unpaired electrons.
- Nearly the same energy.
- Negative charge on the more electronegative atom; positive charge on the electropositive atom.
- Like charges should not reside on adjacent atoms.
Examples:
| Molecule/Ion | No. of Resonance Structures | Feature |
|---|---|---|
| O₃ | 2 | Each O–O bond is intermediate (1.5 order) |
| CO₃²⁻ | 3 | All C–O bonds are equivalent |
| NO₂⁻ | 2 | Both N–O bonds equivalent |
| Benzene (C₆H₆) | 2 | All C–C bonds equivalent |
| SO₄²⁻ | Multiple | Equivalent S–O bonds |
Resonance:
- When a single Lewis structure cannot explain all properties of a compound, two or more structures (canonical forms) are written
- The real molecule does not exist as separate forms, it exists as a resonance hybrid
- Resonance hybrid is a weighted average of all canonical forms
- Due to resonance, electrons are delocalised over the molecule
- Resonance hybrid is more stable (lower energy) than any single structure
Resonance Effect (Mesomeric Effect):
| Type | Explanation | Effect on Electron Density | Examples |
|---|---|---|---|
| +R effect | Group donates electrons to conjugated system | Increases electron density at certain positions | –OH, –OR, –NH₂, –NHR |
| –R effect | Group withdraws electrons from conjugated system | Decreases electron density at o- and p-positions | –NO₂, –CN, –CHO, –COOH |
A temporary effect involving the complete transfer of shared π-electrons to one of the atoms in a multiple bond, in the presence of an attacking reagent.
| Type | Direction of π-electron transfer | Example |
|---|---|---|
| +E Effect | π electrons transferred to the atom to which the reagent gets attached | |
| –E Effect | π electrons transferred to the atom to which the reagent does not attach | 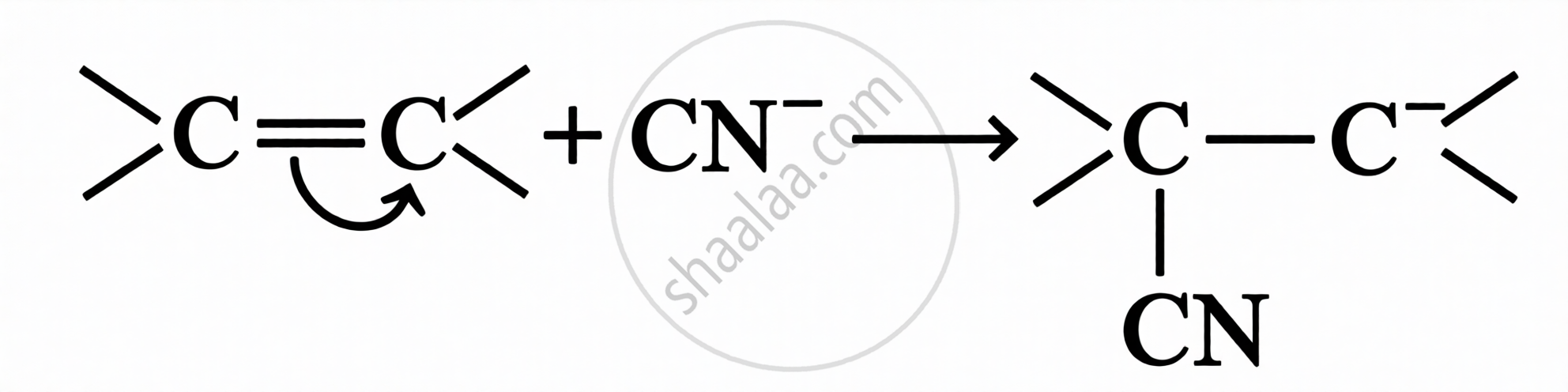 |
Also called the Baker-Nathan effect or no-bond resonance.
Conditions for hyperconjugation:
- The compound must have at least one sp²-hybridised carbon (alkene, alkyl carbocation, or alkyl free radical)
- The α-carbon must have at least one H–C bond attached to the unsaturated system
- More the number of H–C bonds at α-carbon → greater the stabilisation → more stable the alkene/carbocation
Resonance vs. Hyperconjugation:
| Feature | Resonance | Hyperconjugation |
|---|---|---|
| Electrons involved | π electrons delocalised | σ electrons of C–H bond delocalised |
| Stability provided | Greater | Lesser |
| Bond type | π bond | σ bond |
Concepts [21]
- Organic Chemistry
- Structural Representation of Organic Molecules
- Classification of Organic Compounds
- Nomenclature
- IUPAC Names of Straight Chain Alkanes
- IUPAC Names of Branched Saturated Hydrocarbons
- Rules for IUPAC Nomenclature of Branched Saturated Hydrocarbons
- IUPAC Nomenclature of Unsaturated Hydrocarbons (Alkenes and Alkynes)
- IUPAC Names of Simple Monocyclic Hydrocarbons
- IUPAC Nomenclature of Compounds containing one or more Functional Groups
- IUPAC Nomenclature of Substitued Benzene
- Isomerism
- Theoretical Basis of Organic Reactions
- Types of Cleavage of Covalent Bond
- Types of Reagent
- Electronic Effects in Organic Reaction
- Inductive Effect
- Resonance Structures
- Resonance Effect
- Electromeric Effect (E Effect)
- Hyperconjugation