- Specification: Organic chemistry studies carbon compounds (excluding CO₂, carbonates, etc.).
- Sources: Found in plants, animals, coal, petroleum, wood, and by fermentation.
- Lab Synthesis: Many organic compounds are made in labs today.
- Daily Use: Found in soaps, clothes, fuels, medicines, food, etc.
- Importance: Organic compounds are used in almost every part of life.
Definitions [7]
Definition: Organic Compounds
Organic compounds are the compounds of carbon.
Define isomerism.
Compounds having the same molecular formula but different structural formulas are called isomers and the phenomenon is called isomerism.
Define of chain isomerism.
When two or more compounds have a similar molecular formula but are different in the arrangement of carbon atoms in straight or branched chains, the compounds are referred to as chain isomers.
Define a functional group.
An atom or a group of atoms attached to an alkyl group, which takes part in most of the chemical reactions, is called a functional group or functional radical.
Definition: Nomenclature
Nomenclature is the system of assignment of names to organic compounds.
Definition: Isomers
Compounds having the same molecular formula but different structural formula are known as Isomers and the phenomenon is known as Isomerism. (iso = same, meros = parts).
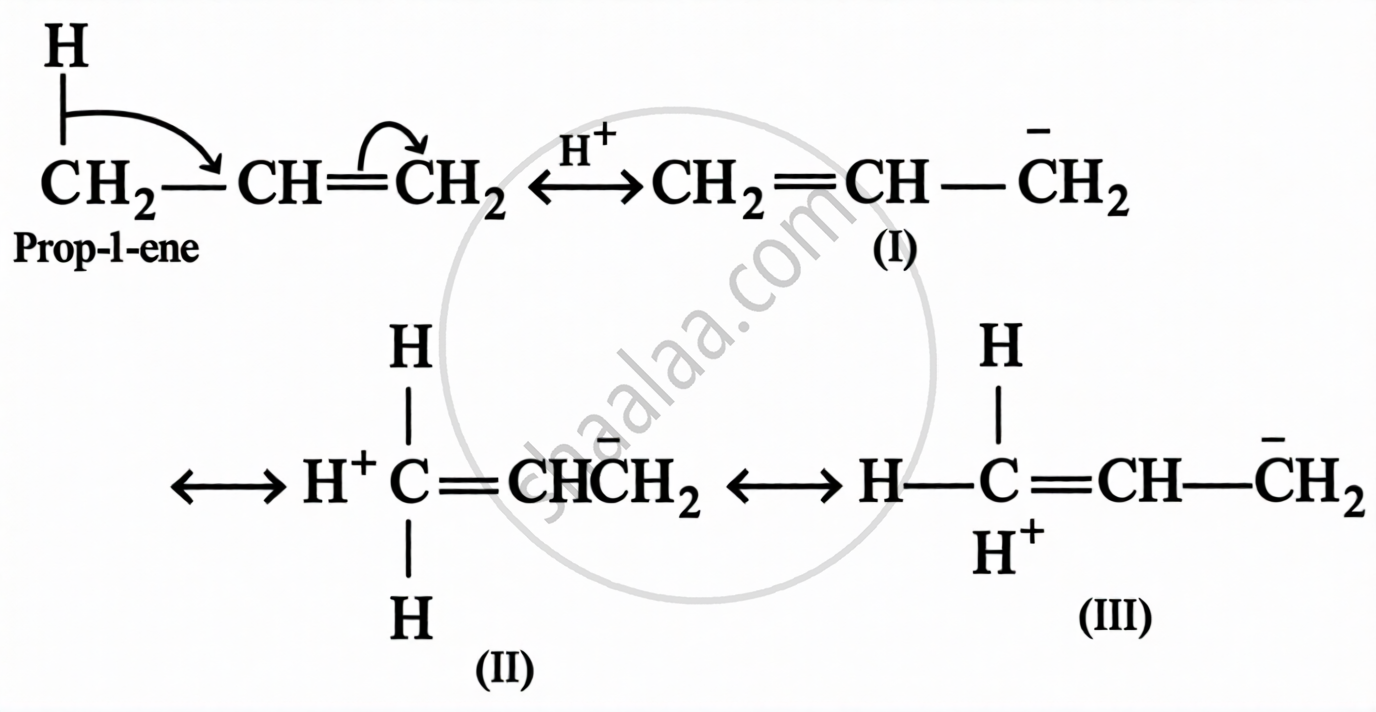
Definition: Hyperconjugation
The stabilising interaction that involves delocalisation of σ-electrons of C–H bond of an alkyl group linked directly to an atom of unsaturated system or to an atom having unshared p-orbital, is called hyperconjugation.
e.g.
Formulae [1]
Give the general formula of carboxylic acids.
CnH2n+1COOH (or RCOOH)
Key Points
Key Points: Tetravalence of Carbon & Shapes of Organic Compounds
Carbon has 4 electrons in its outermost shell → forms 4 covalent bonds → tetravalent.
Determination of Hybridisation (First Method):
- Count the number of pi (π) bonds on that carbon atom.
- Single-bonded C → sp³ hybridisation
- C with one π bond → sp²
- C with two π bonds → sp
| Bonds on C | Hybridisation | Geometry | Example |
|---|---|---|---|
| 4 sigma, 0 pi | sp³ | Tetrahedral | Methane (CH₄) |
| 3 sigma, 1 pi | sp² | Trigonal planar | Ethene (C₂H₄) |
| 2 sigma, 2 pi | sp | Linear | Ethyne (C₂H₂) |
Shapes of molecules:
- Methane (CH₄) → sp³ hybrid orbitals
- Ethene (C₂H₄) → sp² hybrid orbitals
- Ethyne (C₂H₂) → sp hybrid orbitals
Key Points: Organic Compounds
Key Points: Classification of Organic Compounds
(i) Based on Functional Group:
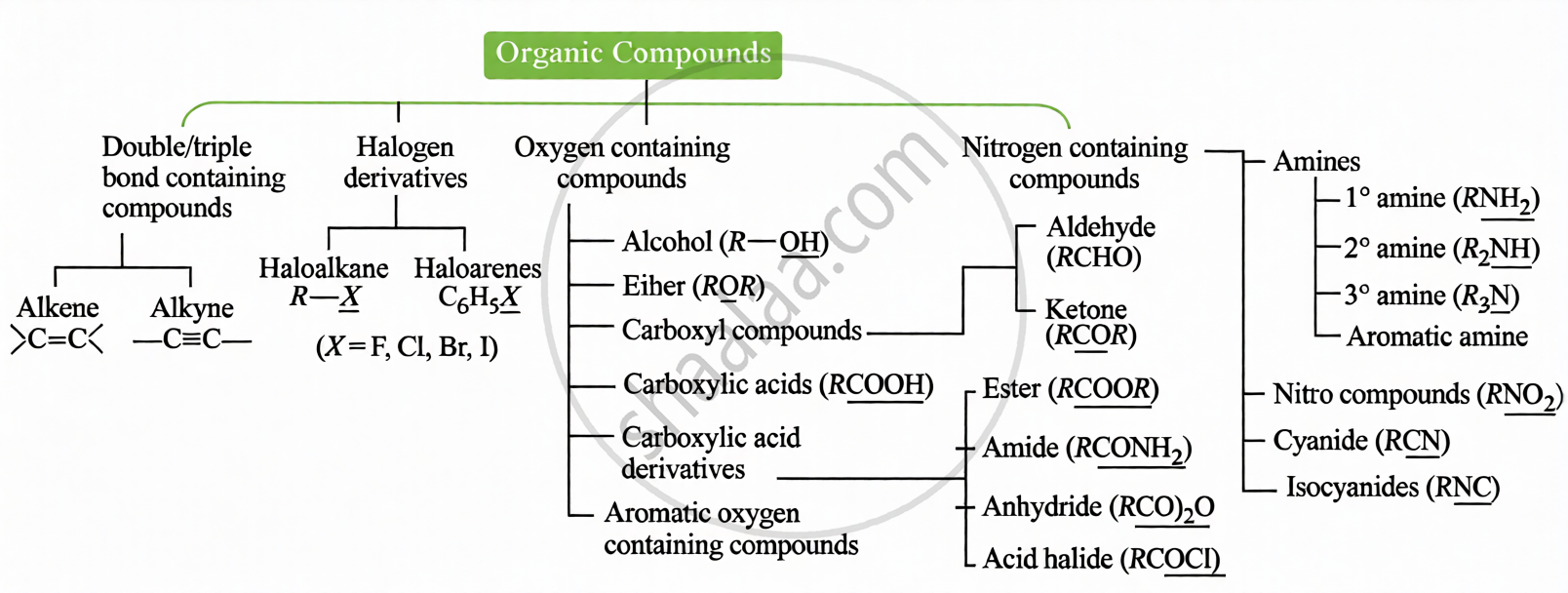
(ii) Based on Carbon Skeleton:
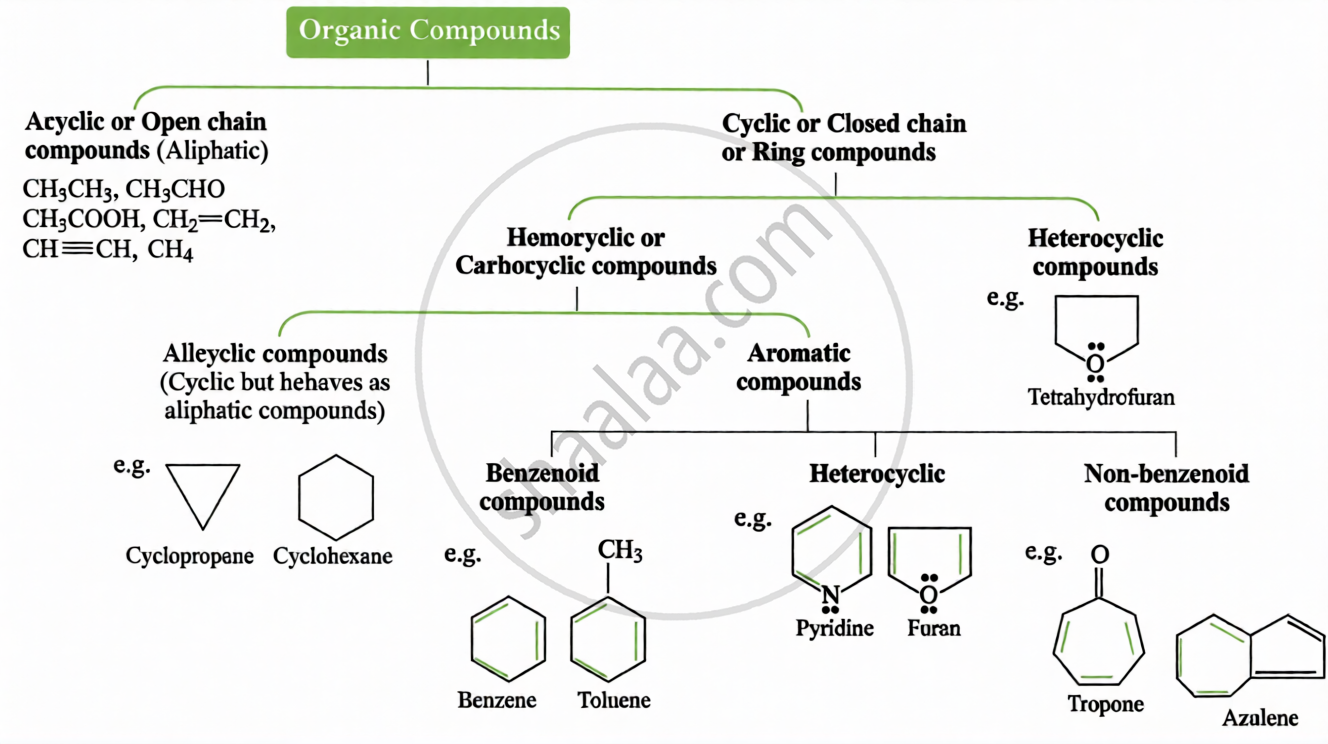
Key points: Nomenclature of Carbon Compounds
- The IUPAC system provides a unique, systematic way to name carbon compounds based on structure, replacing confusing common names.
- An IUPAC name has three parts: prefix, parent, and suffix, reflecting the carbon chain and functional group.
- The parent name is based on the longest carbon chain, and its ending changes to –ane, –ene, or –yne depending on the number of bonds.
- Functional groups are shown as prefixes or suffixes, and the chain is numbered to give them the lowest possible number.
- If the suffix begins with a vowel, the final ‘e’ in the parent alkane name is dropped (e.g., propane → propanone).
Key Points: Types of Isomerism
1. Structural Isomerism
Same molecular formula but different connectivity (bonding) of atoms.
Types:
- Chain isomerism: Different carbon skeleton (straight/branched)
- Position isomerism: Functional group at different positions
- Functional isomerism: Different functional groups (e.g., alcohol vs ether)
- Metamerism: Different alkyl groups on either side of same functional group
- Tautomerism: Dynamic equilibrium between two forms (keto ↔ enol)
2. Stereoisomerism
Same molecular formula and bonding but different spatial arrangement.
Types:
I. Geometrical isomerism:
- Due to restricted rotation (double bond)
- Forms cis (same side) and trans (opposite side)
II. Optical isomerism:
- Mirror image isomers (enantiomers)
- Show optical activity (rotate plane polarized light)
Key Points: Inductive Effect
- Permanent effect due to shift of σ-electrons
- Electrons move towards more electronegative atom
- Effect decreases with distance (negligible after 3 carbons)
e.g-
![]()
| Type | Meaning | Example |
|---|---|---|
| –I effect | Electron withdrawing group | –NO₂, –Cl |
| +I effect | Electron donating group | –CH₃ |
Key Points: Resonance Effect
Resonance:
- When a single Lewis structure cannot explain all properties of a compound, two or more structures (canonical forms) are written
- The real molecule does not exist as separate forms, it exists as a resonance hybrid
- Resonance hybrid is a weighted average of all canonical forms
- Due to resonance, electrons are delocalised over the molecule
- Resonance hybrid is more stable (lower energy) than any single structure
Resonance Effect (Mesomeric Effect):
| Type | Explanation | Effect on Electron Density | Examples |
|---|---|---|---|
| +R effect | Group donates electrons to conjugated system | Increases electron density at certain positions | –OH, –OR, –NH₂, –NHR |
| –R effect | Group withdraws electrons from conjugated system | Decreases electron density at o- and p-positions | –NO₂, –CN, –CHO, –COOH |
Key Points: Electromeric Effect
A temporary effect involving the complete transfer of shared π-electrons to one of the atoms in a multiple bond, in the presence of an attacking reagent.
| Type | Direction of π-electron transfer | Example |
|---|---|---|
| +E Effect | π electrons transferred to the atom to which the reagent gets attached | |
| –E Effect | π electrons transferred to the atom to which the reagent does not attach | 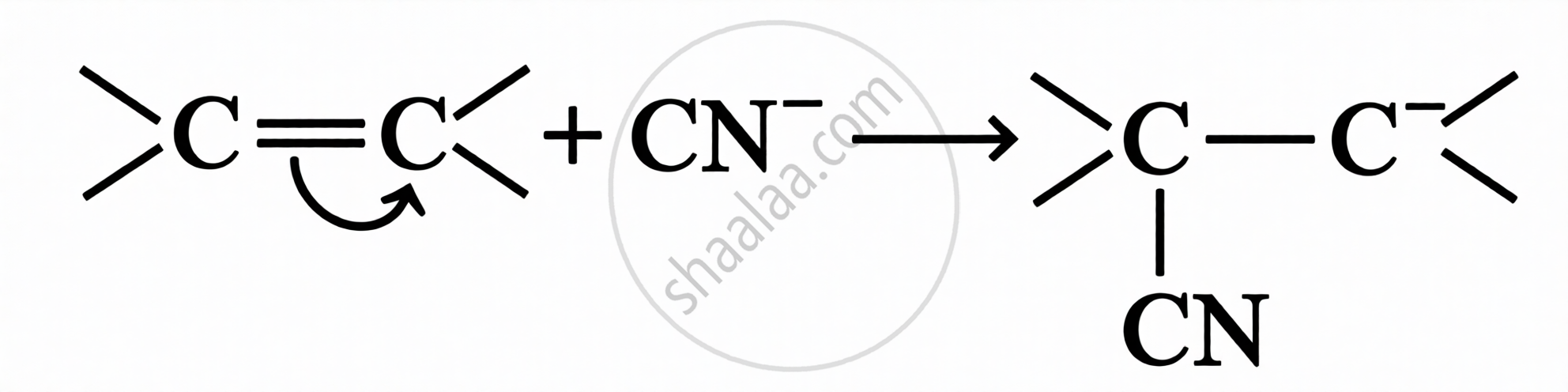 |
Key Points: Hyperconjugation
Also called the Baker-Nathan effect or no-bond resonance.
Conditions for hyperconjugation:
- The compound must have at least one sp²-hybridised carbon (alkene, alkyl carbocation, or alkyl free radical)
- The α-carbon must have at least one H–C bond attached to the unsaturated system
- More the number of H–C bonds at α-carbon → greater the stabilisation → more stable the alkene/carbocation
Resonance vs. Hyperconjugation:
| Feature | Resonance | Hyperconjugation |
|---|---|---|
| Electrons involved | π electrons delocalised | σ electrons of C–H bond delocalised |
| Stability provided | Greater | Lesser |
| Bond type | π bond | σ bond |
Concepts [21]
- Tetravalence of Carbon - Shapes of Organic Compounds
- Organic Compounds
- Complete, Condensed and Bond-line Structural Formulas
- Three-dimensional Representation of Organic Molecules
- Classification of Organic Compounds
- Nomenclature
- IUPAC Nomenclature of Alkanes
- Nomenclature of Organic Compounds having Functional Group(s)
- Nomenclature of Substituted Benzene Compounds
- Isomerism
- Fundamental Concepts in Organic Reaction Mechanism
- Fission of a Covalent Bond
- Nucleophiles and Electrophiles
- Electron Movement in Organic Reactions
- Electron Displacement Effects in Covalent Bonds
- Inductive Effect
- Resonance Structure
- Resonance Effect
- Electromeric Effect (E Effect)
- Hyperconjugation
- Types of Organic Reactions and Mechanisms
