Definitions [14]
Define the term solution.
A homogeneous mixture of two or more substances, whose relative amounts may be changed within certain limits, is called a solution.
Define isotonic solutions.
Two or more solutions exerting the same osmotic pressure are called isotonic solutions.
When two solutions are separated by a semipermeable membrane and no osmosis occurs, i.e., there is no net flow of water on either side through the membrane, the solutions are said to be isotonic solutions. If the membrane is perfectly semipermeable, the two solutions possess the same osmotic pressure and are also referred to as iso-osmotic solutions.
Define the term:
Ideal solution
It is characterised as a solution that adheres to Raoult's Law, with no interactions between the molecules and no volume or heat change during mixing.
For an ideal solution, Enthalpy of mixing of the pure components to form the solution is Δmix H = 0 and the volume of mixing is Δmix V = 0.
A solution which contains more solute than would be necessary to saturate it at a given temperature is called a supersaturated solution.
A solution in which more solute can be dissolved without raising the temperature is called an unsaturated solution.
A solution in which no solute can be dissolved further at a given temperature is called a saturated solution.
Mole fraction of a constituent is the fraction obtained by dividing the number of moles of that constituent by the total number of moles of all the constituents present in the solution.
\[x_{1}=\frac{n_{1}}{n_{1}+n_{2}+n_{3}+...+n_{i}}\]
Define Normality.
Normality (N) of a solution is defined as the number of gram equivalents of the solute present in one liter of the solution. Normality is used in acid-based redox titrations.
Normality (N) = `"Number of gram equivalents of solute"/"Volume of solution in litre"`
Define mole fraction.
The mole fraction of a particular component in a solution is the ratio of the number of moles of that component to the total number of moles of all the components present in the solution.
Define mass percentage.
The mass percentage of a component of a solution is defined as the mass of the solute in grams present in 100 g of the solution.
Define molality.
Molality (m) is defined as the number of moles of the solute dissolved in one kilogram (Kg) of the solvent. The units of molality are moles per kilogram, i.e., mole kg−1. The molality is preferred over molarity if the volume of the solution is either expanding or contracting with temperature.
molality (m) = `"Number of mole of solute"/"mass of solvent (in kg)"`
Define molarity (M).
Molarity (M) is defined as the number of moles of solute dissolved in one litre (or one cubic decimetre) of solution.
Define the term abnormal molar mass.
When the molar mass calculated using colligative properties differs from the theoretical molar mass, it is called an abnormal molar mass.
Define van’t Hoff factor.
The ratio of the observed (experimental) value of a colligative property to the normal (calculated) value of the same property is termed as van’t Hoff factor, i.
Formulae [3]
\[\mathrm{Molarity}=\frac{\text{Number of moles of solute}}{\text{Volume of solution in litres}}\]
\[=\frac{w}{\mathrm{Molar~Mass}}\times\frac{1000}{V}\]
\[\mathrm{Normality}=\frac{\text{Number of gram equivalents of solute}}{\text{Volume of solution of litres}}\]
Normality of a solution = Molarity × nf
\[\mathrm{Molality~}=\frac{\text{Number of moles of solute}}{\text{Mass of solvent in kg}}=\frac{W_{\mathrm{B}}}{M_{\mathrm{B}}}\times\frac{1000}{W_{\mathrm{A}}}\]
where A = solvent, B = solute
Key Points
Based on the physical states of solute and solvent, there are 9 types of solutions:
| S.No. | Solute | Solvent | Example |
|---|---|---|---|
| 1 | Solid | Liquid | Sea water, sugar in water, benzoic acid in benzene |
| 2 | Solid | Solid | Alloys — brass, bronze |
| 3 | Solid | Gas | Iodine in air |
| 4 | Liquid | Liquid | Ethanol in water, gasoline |
| 5 | Liquid | Solid | Amalgam (mercury in silver) |
| 6 | Liquid | Gas | Chloroform in nitrogen |
| 7 | Gas | Liquid | Carbonated water (CO₂ in water), O₂ in water |
| 8 | Gas | Solid | H₂ in palladium |
| 9 | Gas | Gas | Air (O₂, N₂, Ar mixture) |
For ideal solution: ΔHmix = 0, ΔVmix = 0
For non-ideal solution: ΔHmix ≠ 0, ΔVmix ≠ 0
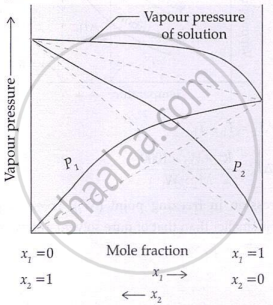
- Positive deviation: A-B interaction < A-A or B-B interactions
- Negative deviation: A-B interaction > A-A or B-B interactions
Plots for Ideal and Non-Ideal Solutions: Formation of ideal solutions can also be represented graphically.
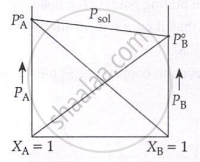
Formation of non-ideal solutions with negative deviation can be represented as:
Azeotropic Mixtures:
A type of liquid mixture having a definite composition and boiling like a pure liquid (i.e., constant boiling mixture).
| Type | Deviation | Example | Boiling Point |
|---|---|---|---|
| Minimum Boiling Azeotrope | Positive deviation | 95.37% (w/w) ethanol + water | Less than either component |
| Maximum Boiling Azeotrope | Negative deviation | 68% (w/w) HNO₃; 20.3% HCl | More than either component |
