Definitions [3]
Definition: Alkanes
Alkanes are hydrocarbons in which all the linkages between the carbon atoms are single covalent bonds.
Definition: Alkenes
Alkenes are unsaturated hydrocarbons containing at least one C=C double bond.
Definition: Alkynes
Alkynes are aliphatic unsaturated hydrocarbons containing at least one C≡C triple bond.
Key Points
Key Points: Alkanes
- General formula: CₙH₂ₙ₊₂ (where n = number of carbon atoms)
- Suffix used for IUPAC naming: –ane
- e.g., Methane (CH₄), Ethane (C₂H₆), Propane (C₃H₈)
- Alkanes exhibit chain isomerism due to absence of any functional group and the possibility of more than one chain type for the same molecular formula
- e.g., C₅H₁₂ forms n-pentane, neo-pentane, and iso-pentane
Key Points: Alkenes
- General formula: CₙH₂ₙ (where n = 2, 3, 4…)
- Suffix for IUPAC naming: –ene
- e.g., Ethene (CH₂=CH₂), Propene (CH₃–CH=CH₂)
- The double bond consists of one σ bond and one π bond
Key Points: Alkynes
- General formula: CₙH₂ₙ₋₂
- Suffix for IUPAC naming: –yne
- e.g., Propyne (CH₃–C≡CH), Butyne (CH₃–CH₂–C≡CH)
- C₂H₂ is acetylene (common name); IUPAC name is ethyne
- The triple bond consists of one σ bond and two π bonds
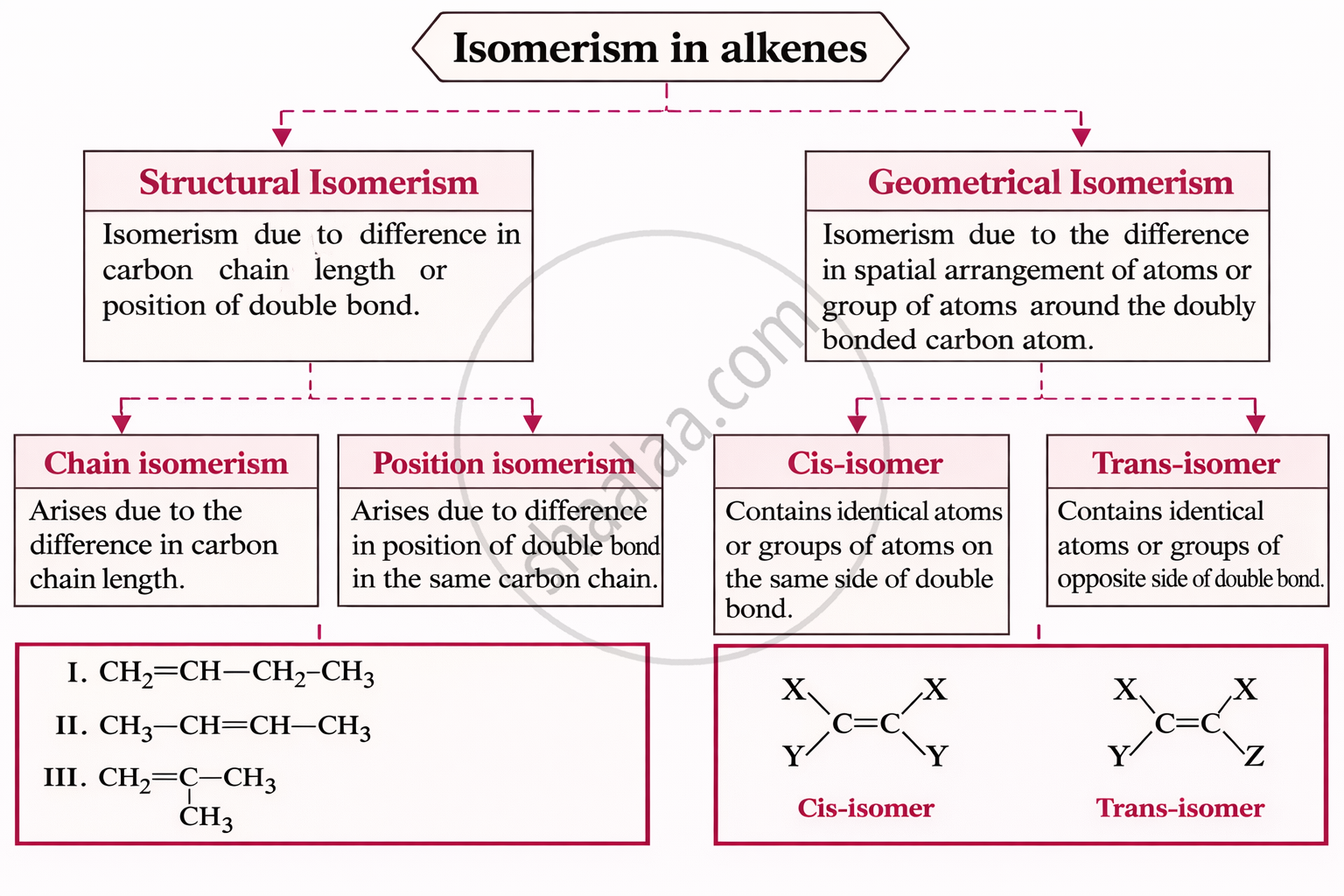
Isomerism in Alkynes:
- Alkynes show position isomerism (type of structural isomerism)
- e.g., 1-Butyne and 2-Butyne
Key Points: Aromatic Hydrocarbons
Cyclic, planar hydrocarbons with delocalised π electrons.
Benzenoids contain benzene ring; Non-benzenoids are aromatic without benzene ring.
Structure of Benzene:
- All 6 C atoms are sp² hybridised
- Unhybridised p-orbitals overlap laterally → delocalised π bonds
- Bond length = 139 pm (uniform, due to resonance)
Huckel's Rule
- Cyclic and planar
- Each ring atom has a p-orbital
- Contains (4n + 2) π electrons (n = 0, 1, 2…)
Examples: Benzene, Naphthalene, Pyridine.
Physical Properties:
- Colourless liquid, sweet smell
- BP = 353 K, MP = 278.5 K
- Immiscible with water; burns with sooty flame
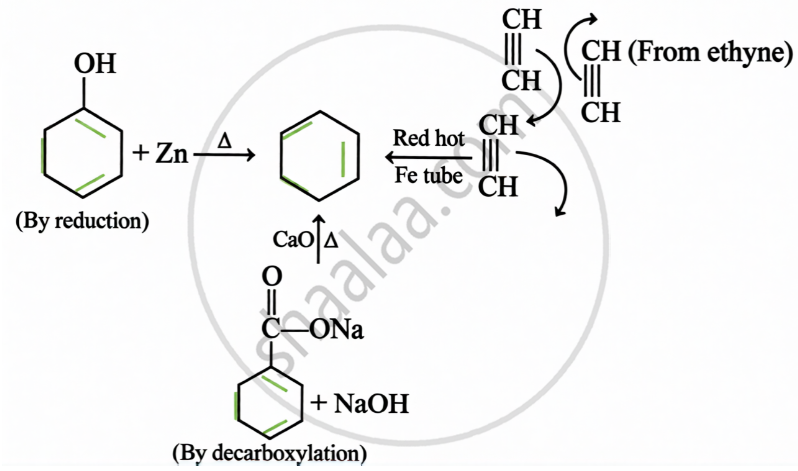
Preparation:
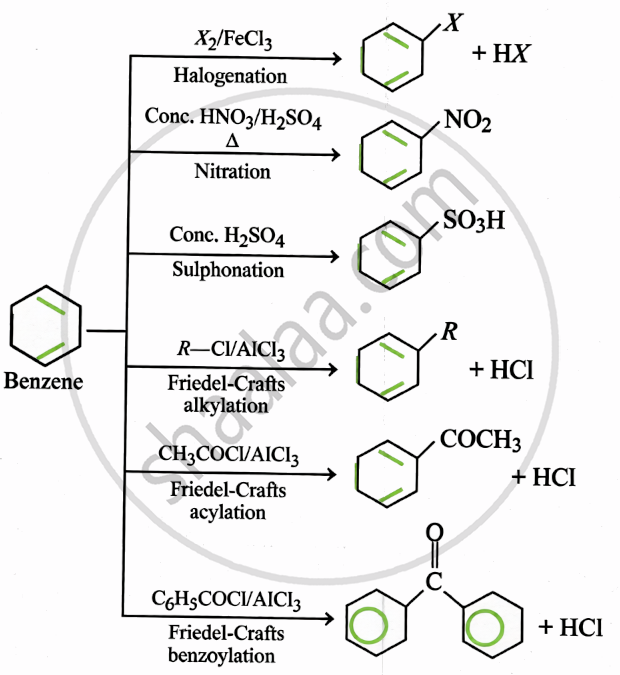
Electrophilic Substitution: