Definitions [2]
Define transition metals.
Elements having partially filled d-orbitals in ground state or in excited state are known as transition elements.
Define lanthanoid contraction.
In the lanthanoids, the electrons are filling the 4f-subshell. On moving from left to right, the nuclear charge increases, and this increase is expected to be compensated by the increase in the magnitude of the shielding effect by the 4f-electrons. However, the f-electrons have very poor shielding effects. With an increasing atomic number in the lanthanoid series, there is a progressive decrease in the atomic as well as ionic radii of trivalent ions from La3+ to Lu3+, and this is known as lanthanoid contraction.
Key Points
d-Block Elements:
- Elements in which the last electron enters (n−1)d orbital.
- Located in the middle of the periodic table.
- Also called transition elements.
- Show variable oxidation states, colored compounds, complex formation.
General Electronic Configuration: \[(n-1)d^{1-10}ns^{0-2}\]
f-Block Elements:
- Elements in which the last electron enters (n−2)f orbital.
- Placed separately at the bottom of the periodic table.
- Known as inner transition elements.
Two series:
- Lanthanides (4f): Ce (58) → Lu (71)
- Actinides (5f): Th (90) → Lr (103)
General Electronic Configuration: \[(n-2)f^{1-14}(n-1)d^{0-1}ns^{2}\]
- Located between s-block and p-block elements
- Occupy Groups 3 to 12
- Present in 4 periods (4th to 7th)
Series of d-block:
| Series | Elements |
|---|---|
| 3d | Sc (21) → Zn (30) |
| 4d | Y (39) → Cd (48) |
| 5d | La (57) → Hg (80) |
| 6d | Ac (89) → Cn (112) |
Electronic Configuration:
- The general electronic configuration of 3d-series elements is \[3d^{1-10}4s^{1-2}\].
- The general electronic configuration of 4d-series elements is \[4d^{1-10}5s^{1-2}\].
- The general electronic configuration of 5d-series elements is \[5d^{1-10}6s^{1-2}\].
- The general electronic configuration of 6d-series elements is \[6d^{1-10}7s^{1-2}\].
- All transition elements are d-block elements, but all d-block elements are not transition elements. Zn, Cd and Hg are not transition elements.
| 3d-Series | At. No | Outer E.C | 4d-Series | At. No | Outer E.C | 5d-Series | At. No | Outer E.C | 6d-Series | At. No | Outer E.C |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Sc | 21 | 3d¹4s² | Y | 39 | 4d¹5s² | La | 57 | 5d¹6s² | Ac | 89 | 6d¹7s² |
| Ti | 22 | 3d²4s² | Zr | 40 | 4d²5s² | Hf | 72 | 5d²6s² | Rf | 104 | 6d²7s² |
| V | 23 | 3d³4s² | Nb | 41 | 4d⁴5s¹ | Ta | 73 | 5d³6s² | Db | 105 | 6d³7s² |
| Cr | 24 | 3d⁵4s¹ | Mo | 42 | 4d⁵5s¹ | W | 74 | 5d⁴6s² | Sg | 106 | 6d⁴7s² |
| Mn | 25 | 3d⁵4s² | Tc | 43 | 4d⁵5s² | Re | 75 | 5d⁵6s² | Bh | 107 | 6d⁵7s² |
| Fe | 26 | 3d⁶4s² | Ru | 44 | 4d⁷5s¹ | Os | 76 | 5d⁶6s² | Hs | 108 | 6d⁶7s² |
| Co | 27 | 3d⁷4s² | Rh | 45 | 4d⁸5s¹ | Ir | 77 | 5d⁷6s² | Mt | 109 | 6d⁷7s² |
| Ni | 28 | 3d⁸4s² | Pd | 46 | 4d¹⁰5s⁰ | Pt | 78 | 5d⁹6s¹ | Ds | 110 | 6d⁸7s² |
| Cu | 29 | 3d¹⁰4s¹ | Ag | 47 | 4d¹⁰5s¹ | Au | 79 | 5d¹⁰6s¹ | Rg | 111 | 6d¹⁰7s¹ |
| Zn | 30 | 3d¹⁰4s² | Cd | 48 | 4d¹⁰5s² | Hg | 80 | 5d¹⁰6s² | Cn | 112 | 6d¹⁰7s² |
Atomic and Ionic Radii:
- Atomic and ionic radii of d-block elements are smaller than s-block but larger than p-block elements.
- Within a 3d series, atomic radii decrease for the first five elements (Sc to Mn), then remain almost constant for the next five (Fe to Zn). This is because the increase in ENC (effective nuclear charge) first causes shrinkage, but additional d-electrons increase shielding and counterbalance further shrinkage.
- The 4d and 5d series elements have larger atomic and ionic radii than 3d series elements (due to more electron shells). However, 4d and 5d elements have nearly the same size — due to lanthanoid contraction.
Atomic Volume and Density:
- Atomic volume decreases along a period (as atomic size decreases).
- Density increases along the period.
| Element | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn |
|---|---|---|---|---|---|---|---|---|---|---|
| Density (g/cm³) | 3.43 | 4.1 | 6.07 | 7.19 | 7.21 | 7.8 | 8.7 | 8.9 | 8.9 | 7.1 |
Melting and Boiling Points:
- All transition elements have high melting points (typically above 900°C) in their solid state.
- Zn, Cd, Hg have abnormally low melting points because their completely filled d-orbitals prevent strong covalent metallic bonding.
- As unpaired electrons increase, metallic bonding strengthens → higher melting point. Tungsten (W) has the highest melting point of all metals.
- Mn and Tc have abnormally low melting points.
Enthalpies of Atomisation:
- Due to strong interatomic attraction, transition metals have high enthalpies of atomisation.
- Greater the number of valence electrons → stronger metallic bonding → higher enthalpy of atomisation.
- Members of 4d and 5d series have greater enthalpy of atomisation than 3d series.
Ionisation Energies:
- IE values of d-block elements lie between those of s-block and p-block elements.
- IE first increases up to Mn, then becomes irregular or constant due to the irregular trend of atomic size in 3d series.
- IE of Zn, Cd, and Hg are abnormally high due to the greater stability of completely filled d-subshells.
- The first two IE values of Ni are lower than Pt → Ni(II) compounds are more thermodynamically stable than Pt(II).
IE₁ order (important anomalies):
- Hg > Cd > Zn
- Au > Cu > Ag
- Pt > Pd > Ni
Oxidation States:
All transition elements except the first and last of each series show a number (variable) of oxidation states.
| Element | Oxidation States |
|---|---|
| Sc | +3 |
| Ti | +2, +3, +4 |
| V | +2, +3, +4, +5 |
| Cr | +2, +3, +4, +5, +6 |
| Mn | +2, +3, +4, +5, +6, +7 |
| Fe | +2, +3, +4, +6 |
| Co | +2, +3, +4 |
| Ni | +2, +3, +4 |
| Cu | +1, +2 |
| Zn | +2 |
- Mn shows the maximum number of oxidation states in the first series (7 states) — because it has 5 unpaired 3d electrons + 2 s-electrons available.
- Higher oxidation states are more stable for heavier members of a group (e.g., Mo(VI) and W(VI) are more stable than Cr(VI)).
- Lower oxidation states are more stable for lighter (3d) members.
Standard Electrode Potential:
- No regular trend exists in E° (M²⁺/M) values because IE and sublimation enthalpies show irregular variation.
- SRP tends to become more positive across a period (left to right) due to increasing IE and decreasing atomic size.
- Within a group, SRP becomes more negative going down.
| E°/V | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn |
|---|---|---|---|---|---|---|---|---|---|---|
| M²⁺/M | — | −1.63 | −1.18 | −0.90 | −1.18 | −0.44 | −0.28 | −0.25 | +0.34 | −0.76 |
| M³⁺/M²⁺ | — | −0.37 | −0.26 | −0.41 | +1.57 | +0.77 | +1.97 | — | — | — |
- E° for Ni²⁺/Ni and Zn²⁺/Zn are more negative than expected. The high negative value of Ni²⁺/Ni stabilises Ni²⁺ ions. The high negative value for Zn²⁺/Zn is due to the stable, completely filled 3d¹⁰ configuration.
- Cr²⁺ is a strong reducing agent (acts as a reducing agent, gets oxidised to Cr³⁺; the d³ configuration = t₂g³ is very stable).
- Mn³⁺ (d⁴) is an oxidising agent — it gets reduced to Mn²⁺ (d⁵), which has an exactly half-filled d-orbital (extra stability).
- E°(Mn²⁺/Mn) is more negative than expected — due to extra stability of the half-filled 3d⁵ (Mn²⁺) ion.
Coloured Ions: Most of the transition metal compounds (ionic as well as covalent) are coloured both in the solid and in aqueous solution, in contrast to the compounds of s and p-block elements.
| Ion | Configuration | Colour |
|---|---|---|
| Sc³⁺ | 3d⁰ | Colourless |
| Ti⁴⁺ | 3d⁰ | Colourless |
| Ti³⁺ | 3d¹ | Purple |
| V⁴⁺ | 3d¹ | Blue |
| V³⁺ | 3d² | Green |
| V²⁺ | 3d³ | Violet |
| Cr³⁺ | 3d³ | Violet |
| Mn³⁺ | 3d⁴ | Violet |
| Cr²⁺ | 3d⁴ | Blue |
| Mn²⁺ | 3d⁵ | Pink |
| Fe³⁺ | 3d⁵ | Yellow |
| Fe²⁺ | 3d⁶ | Green |
| Co³⁺ | 3d⁶ | Blue |
| Co²⁺ | 3d⁷ | Pink |
| Ni²⁺ | 3d⁸ | Green |
| Cu²⁺ | 3d⁹ | Blue |
| Zn²⁺ | 3d¹⁰ | Colourless |
Magnetic Properties: In the case of transition metals, as they contain unpaired electrons in (n – 1)d orbitals, most of the transition metal ions and their compounds are paramagnetic.
Magnetic moment is calculated by spin only formula viz.
\[\mu=\sqrt{n\left(n+2\right)}\mathrm{~B.M.}\]
where n = number of unpaired electrons
I. K₂Cr₂O₇ (Potassium dichromate)
Preparation: From chromite (FeO·Cr₂O₃)
4FeO·Cr₂O₃ + O₂ → 2Fe₂O₃ + 4Cr₂O₃
4Na₂CO₃ + 2Cr₂O₃ + 3O₂ → 4Na₂Cr₂O₄ + 4CO₂ × 2
4FeO·Cr₂O₃ + 8Na₂CO₃ + 7O₂ → 8Na₂Cr₂O₄ + 2Fe₂O₃ + 8CO₂
2Na₂CrO₄ + 8H₂SO₄ → Na₂Cr₂O₇ + Na₂SO₄ + H₂O
Na₂Cr₂O₇ + 2KCl → K₂Cr₂O₇ + 2NaCl
Properties
Oxidising Properties:
- Liberates I₂ from KI
K₂Cr₂O₇ + 7H₂SO₄ + 6KI → 4K₂SO₄ + Cr₂(SO₄)₃ + 3I₂ + 7H₂O - Oxidises ferrous salts to ferric salts:
K₂Cr₂O₇ + 7H₂SO₄ + 6FeSO₄ → K₂SO₄ + Cr₂(SO₄)₃ + 3Fe₂(SO₄)₃ + 2H₂O - Oxidises H₂S to sulphur:
K₂Cr₂O₇ + 4H₂SO₄ + 3H₂S → K₂SO₄ + Cr₂(SO₄)₃ + 7H₂O + 3S - Oxidises sulphites to sulphates:
K₂Cr₂O₇ + 4H₂SO₄ + 3Na₂SO₃ → K₂SO₄ + Cr₂(SO₄)₃ + 4H₂O + 3Na₂SO₄ - Oxidises nitrites to nitrates:
K₂Cr₂O₇ + 4H₂SO₄ + 3NaNO₂ → K₂SO₄ + Cr₂(SO₄)₃ + 3NaNO₃ + 4H₂O
Structures:
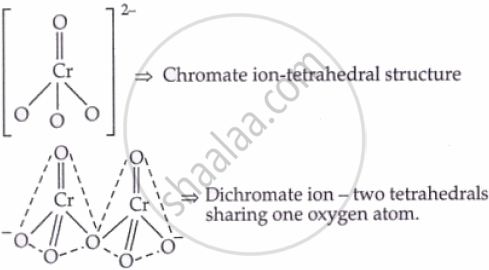
II. Potassium Permanganate (KMnO₄)
Preparation: From Pyrolusite (MnO₂)
2MnO₂ + 4KOH + O₂ → 2K₂MnO₄ + 2H₂O
2MnO₂ + 2K₂CO₃ + O₂ → 2K₂MnO₄ + 2CO₂
MnO₂ + 2KOH + KNO₃ → K₂MnO₄ + KNO₂ + H₂O
3K₂MnO₄ + 2CO₂ → 2KMnO₄ + MnO₂↓ + 2K₂CO₃
Properties
Oxidising Properties:
- Oxidising H₂S to S
2KMnO₄ + 3H₂SO₄ + 5H₂S → K₂SO₄ + 2MnSO₄ + 3H₂O + 5S - Oxidises sulphur dioxide to sulphuric acid
2KMnO₄ + 5SO₂ + 2H₂O → K₂SO₄ + 2MnSO₄ + 2H₂SO₄ - Oxidises oxalates to CO₂
2KMnO₄ + 3H₂SO₄ + 5C₂H₂O₄ → K₂SO₄ + 2MnSO₄ + 8H₂O + 10CO₂ - Oxidises HX to X₂, where X = Cl, Br, I
2KMnO₄ + 3H₂SO₄ + 10HX → K₂SO₄ + 2MnSO₄ + 8H₂O + 5X₂
Structure:
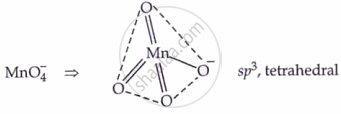
Position and Introduction:
The f-block consists of two series:
- Lanthanoids — fourteen elements from Ce (58) to Lu (71), following Lanthanum (La, 57)
- Actinoids — fourteen elements from Th (90) to Lr (103), following Actinium (Ac, 89)
In lanthanides, electrons enter the penultimate (4f) and pre-penultimate subshells.
General configuration of lanthanoids: [Xe] 4f¹⁻¹⁴ 5d⁰⁻¹ 6s²
Complete Lanthanoid Table:
| Element | Symbol | Z | Outer Config. | Oxidation States | M³⁺ radius (pm) |
|---|---|---|---|---|---|
| Lanthanum | La | 57 | 5d¹, 6s² | +3 | 106 |
| Cerium | Ce | 58 | 4f¹, 5d¹, 6s² | +3, +4 | 103 |
| Praseodymium | Pr | 59 | 4f³, 6s² | +3, +4 | 101 |
| Neodymium | Nd | 60 | 4f⁴, 6s² | +2, +3, +4 | 99 |
| Promethium | Pm | 61 | 4f⁵, 6s² | +3 | 98 |
| Samarium | Sm | 62 | 4f⁶, 6s² | +2, +3 | 96 |
| Europium | Eu | 63 | 4f⁷, 6s² | +2, +3 | 95 |
| Gadolinium | Gd | 64 | 4f⁷, 5d¹, 6s² | +3 | 94 |
| Terbium | Tb | 65 | 4f⁹, 6s² | +3, +4 | 92 |
| Dysprosium | Dy | 66 | 4f¹⁰, 6s² | +3, +4 | 91 |
| Holmium | Ho | 67 | 4f¹¹, 6s² | +3 | 89 |
| Erbium | Er | 68 | 4f¹², 6s² | +3 | 88 |
| Thulium | Tm | 69 | 4f¹³, 6s² | +2, +3 | 87 |
| Ytterbium | Yb | 70 | 4f¹⁴, 6s² | +2, +3 | 86 |
| Lutetium | Lu | 71 | 4f¹⁴, 5d¹, 6s² | +3 | 85 |
La (5d¹6s²), Gd (4f⁷5d¹6s²), and Lu (4f¹⁴5d¹6s²) have a 5d¹ electron — they fill 5d before filling 4f again, due to the stability of half-filled (4f⁷) configuration.
Physical State:
- All are silvery white metals with tensile strength; good conductors of heat and electricity.
- Density ranges from 6.77 to 9.74 g/cm³ and increases with atomic number.
- They readily form alloys with other metals, especially iron.
Oxidation States
- Most common and stable OS = +3
- Some exist in +2 (Sm²⁺, Eu²⁺, Tm²⁺, Yb²⁺) — because they achieve stability trying to reach +3 OS.
- Some exist in +4 (Ce⁴⁺, Pr⁴⁺, Tb⁴⁺, Dy⁴⁺) — because they try to approach +3 from +4; hence, these are good oxidising agents.
- Elements in +2 OS act as reducing agents; in +4 OS act as oxidising agents.
Chemical Behaviour:
Lanthanoids (Ln) react with:
| Reagent | Product |
|---|---|
| Halogens | LnX₃ |
| Carbon (2773 K) | LnC₂ |
| Dilute acids | H₂ gas liberated |
| O₂ | Ln₂O₃ |
| N₂ (Δ) | LnN |
| Sulphur | Ln₂S₃ |
| H₂O | Ln(OH)₃ + H₂ |
- Lanthanoids react with boiling water to give a mixture of oxide and hydride.
- They combine with most non-metals at moderate temperatures.
- Alkalis have no action on them.
Actinoids are the 14 elements from Th (90) to Lr (103). General configuration: [Rn] 5f¹⁻¹⁴ 6d⁰⁻² 7s²
Complete Actinoid Table:
| Element | Symbol | Z | E.C. | Oxidation States | M³⁺ radius (pm) | M⁴⁺ radius (pm) |
|---|---|---|---|---|---|---|
| Actinium | Ac | 89 | [Rn] 6d¹ 7s² | +3 | 111 | — |
| Thorium | Th | 90 | [Rn] 6d² 7s² | (+3), +4 | — | 99 |
| Protactinium | Pa | 91 | [Rn] 5f² 6d¹ 7s² | +3, +4, +5 | 96 | — |
| Uranium | U | 92 | [Rn] 5f³ 6d¹ 7s² | +3, +4, +5, +6 | 103 | 93 |
| Neptunium | Np | 93 | [Rn] 5f⁴ 6d¹ 7s² | +3, +4, +5, +6, +7 | 101 | 92 |
| Plutonium | Pu | 94 | [Rn] 5f⁶ 7s² | +3, +4, +5, +6, +7 | 100 | 90 |
| Americium | Am | 95 | [Rn] 5f⁷ 7s² | +3, (+4), +5, +6 | 99 | 89 |
| Curium | Cm | 96 | [Rn] 5f⁷ 6d¹ 7s² | +3, (+4) | 99 | 88 |
| Berkelium | Bk | 97 | [Rn] 5f⁹ 7s² | +3, +4 | 98 | 87 |
| Californium | Cf | 98 | [Rn] 5f¹⁰ 7s² | +3 | — | 86 |
| Einsteinium | Es | 99 | [Rn] 5f¹¹ 7s² | +3 | — | — |
| Fermium | Fm | 100 | [Rn] 5f¹² 7s² | +3 | — | — |
| Mendelevium | Md | 101 | [Rn] 5f¹³ 7s² | +3 | — | — |
| Nobelium | No | 102 | [Rn] 5f¹⁴ 7s² | +3 | — | — |
| Lawrencium | Lr | 103 | [Rn] 5f¹⁴ 6d¹ 7s² | +3 | — | — |
Concepts [9]
- Transition and Inner Transition Elements
- Position in the Periodic Table - d-block Elements
- Electronic Configurations of the d-block Elements
- Physical Properties of the Transition Elements (d-block)
- Important Compounds of Transition Elements
- F-block Elements
- The Lanthanoids
- The Actinoids
- Applications of d and f Block Elements
