Advertisements
Advertisements
Question
Write type of hybridization and magnetic behavior of the following:
[Co(NH3)6]3⊕
Very Short Answer
Advertisements
Solution
[Co(NH3)6]3⊕ has d2sp3 hybridisation.
CO3+ [Ar] 3d6

Low spin complex:
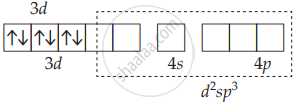
No unpaired electrons: Diamagnetic
shaalaa.com
Is there an error in this question or solution?
2024-2025 (July) Official Board Paper
