Advertisements
Advertisements
Questions
Write the structures of any two chelating ligands.
Illustrate chelating ligands with a suitable example.
Long Answer
Advertisements
Solution
- Ethylenediamine (H2N–CH2–CH2–NH2): It is a bidentate ligand, meaning it has two nitrogen atoms, each with a lone pair, that can bind to metal ions.
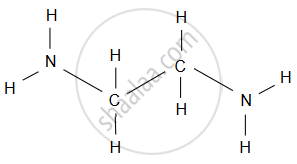
The two nitrogen (N) atoms are the donor atoms that can coordinate to the central metal ion. - Oxalate Ion (\[\ce{C2O^2-_4}\]): The oxalate ion consists of two carboxylate groups (–COO) linked by a central oxygen atom. The oxygen atom is a shared donor atom in the structure.
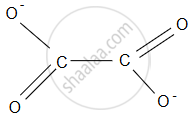
The two oxygen atoms (one from each carboxylate group) can coordinate to a central metal ion, forming a stable chelate ring.
shaalaa.com
Is there an error in this question or solution?
