Advertisements
Advertisements
Questions
Write the structure of the geometrical isomer of the following complex:
[Pt(gly)2]
Write the structure of cis- and trans-isomers of the following complex:
[Pt(gly)2]
Chemical Equations/Structures
Advertisements
Solution
The complex [Pt(gly)2] consists of platinum (Pt) at its center surrounded by two glycinate (gly) ligands, each of which can bind via its amine group (–NH2) and carboxylate group (–COO–). Since glycinate is a bidentate ligand, it attaches to the platinum at two sites. The coordination geometry around platinum (Pt) is typically square planar for a Pt2+ complex.
- Cis isomer: The two glycinate ligands are adjacent to each other (90° angle) in the square planar geometry.
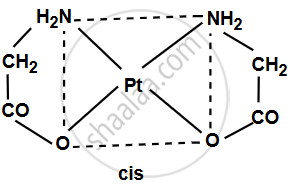
- Trans isomer: The two glycinate ligands are opposite each other (180° angle) in the square planar geometry.
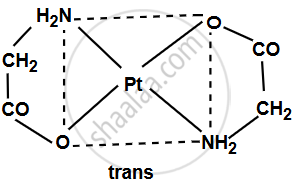
shaalaa.com
Is there an error in this question or solution?
