Advertisements
Advertisements
Question
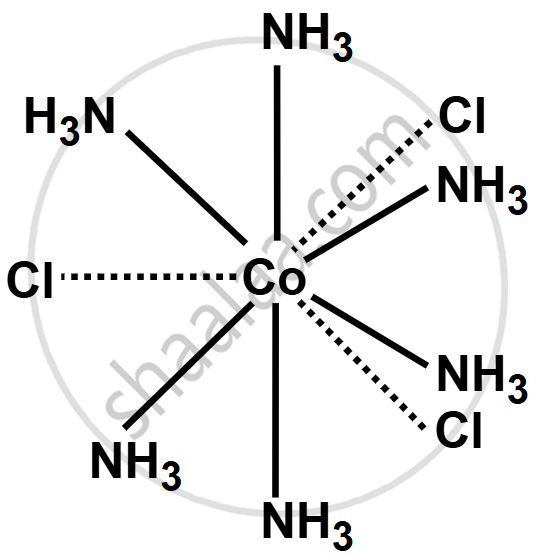
Write the structure of the following complex on the basis of Werner’s theory:
CoCl3·6NH3
Chemical Equations/Structures
Advertisements
Solution
In the complex CoCl3·6NH3, cobalt is in the +3 oxidation state, so its primary valency is 3. The three Cl− ions are linked through primary valency. The coordination number of cobalt is 6, so it forms six secondary valencies with six NH3 molecules. This gives the octahedral structure, with three chloride ions and six ammonia molecules around the central cobalt ion.
shaalaa.com
Is there an error in this question or solution?
