Advertisements
Advertisements
Question
Write the reactions to bring about the following conversions.
Nitroalkane to Alkylamine
Advertisements
Solution
\[\ce{\underset{\text{Nitroalkane}}{R - NO2} + 6[H] ->[Sn/HCl] \underset{\text{Alkylamine}}{R - NH2} + 2H2O}\]
APPEARS IN
RELATED QUESTIONS
Write a short note on Hofmann elimination
Which one of the following compounds does not react with acetyl chloride?
Answer the following
What is diazotisation?
Answer the following
Explain carbylamine reaction with suitable examples.
Write a reaction to convert methanamine into ethanamine.
Answer the following
Write a reaction to convert ethylamine into methylamine.
Write the name of the reaction in which aromatic primary amines produce offensive smell.
Write the reactions to bring about the following conversions.
Methyl chloride to Ethanamine
\[\ce{X ->[Sn/HCl] Propan-2-amine}\]
Compound X is ____________.
Which of the following will NOT give positive carbylamine test?
Identify the amine formed when ethyltrimethyl ammonium iodide is treated with silver hydroxide and further heated strongly.
Select the CORRECT option about the following reaction.
\[\ce{CH3 - CH2 - N^+(CH3)3 - I^- ->[Moist][Ag2O] X ->[\Delta] Y + Z + H2O}\]
Which one of the following compounds when heated with KOH and CHCl3 gives carbylamine test?
Which group from following decreases the basic strength in substituted aniline?
Which one of the following methods is NOT used for the synthesis of amines?
Identify reagent 'A' in the following conversion?
\[\ce{N-N Dimethyl aniline ->[A][273-278 K] 4-nitroso-N-N-dimethylaniline}\]
When propanamide is treated with bromine and alcoholic NaOH, it gives ______.
What is the product obtained in the following reaction?

Which of the following amines liberates nitrogen gas when treated with nitrous acid?
What is the product obtained when ethyl amine is heated with large excess of ethyl iodide?
Select the compound which on treatment with nitrous acid liberates nitrogen.
Identify the X in the reaction.
\[\ce{R - NH2 + HNO2 ->[NaNO2 + HCl][273 - 278 K] X ->[H2O] R - OH + N2\uparrow + HCl}\]
Convert Methyl iodide to methyl isocyanide
Which from following amines forms corresponding alcohol liberating dinitrogen when treated with mixture of NaNO2(aq) and HCI?
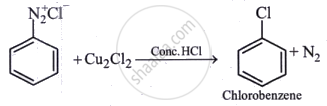
Above reaction is known as ______.
