Advertisements
Advertisements
Questions
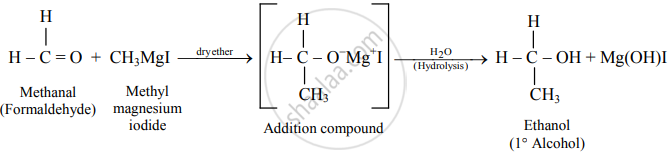
Write the preparation of ethanol from methyl magnesium iodide.
Prepare the following by using methyl magnesium iodide:
Ethanol
Advertisements
Solution
Methyl magnesium iodide reacts with formaldehyde to form an adduct, which on hydrolysis with dilute acid gives ethanol.
APPEARS IN
RELATED QUESTIONS
Anisole on heating with concentrated HI gives ______.
Choose the correct option.
Ethers react with cold and concentrated H2SO4 to form ________.
What is the action of atmospheric oxygen on ethers?
Write the reaction between ethanol and acetic anhydride.
Explain the fact that in alkyl aryl ethers, the alkoxy group is ring activating and ortho/para directing towards electrophilic aromatic substitution.
What are the limitations to prepare ether by this method?
What is the action of following on diethyl ether?
dil. H2SO4
What is the action of following on diethyl ether?
PCl5
In the following reaction:
\[\ce{Ether ->[Hot HI] A + B + H2O}\],
If A and B are identical, the ether is ____________.
For the preparation of mixed ethers by Williamson synthesis, which of the following combination will give the best yield?
Action of hydrogen iodide on anisole gives ______.
____________ on heating with excess of conc. HI gives two moles of ethyl iodide.
In ethers, two C-O sigma bonds are formed by ____________ overlap.
A mixed ether on hydrolysis gives ____________.
\[\ce{(CH3)2CH - O - CH3 ->[Cold HI] X + Y}\]
\[\ce{X ->[K2Cr2O7][dil. H2SO4] Z}\]
\[\ce{Y ->[NaOH(aq)][\Delta] CH3OH}\]
Identify X, Y and Z.
Ether is obtained from ethyl alcohol in presence of H2SO4 at _______.
The major product of the reaction between tert-butyl chloride and sodium ethoxide is ______.
Formation of diethyl ether from ethanol is based oh a ______.
Ethers when dissolved in cold concentrated sulphuric acid forms ______.
Explain Williamson's synthesis.
In Williamson synthesis, if a tertiary alkyl halide is used, then ______.
