Advertisements
Advertisements
Question
Write the names of isotopes of hydrogen.
Short/Brief Note
Advertisements
Solution
Hydrogen has three naturally occuring isotopes namely Protium (1H1), Deuterium (1H2) and Tritium (1H3).
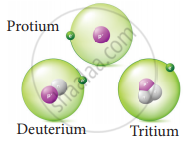
Isotopes of hydrogen
shaalaa.com
Is there an error in this question or solution?
Chapter 4: Hydrogen - Evaluation [Page 123]
