Advertisements
Advertisements
Question
Write the molecular formula and possible structural formula of the first four members of homologous series of carboxylic acids.
Advertisements
Solution
| S.No. | Name | Molecular formula | Structural formula |
| 1. | Formic acid | CH2O2 | \[\begin{array}{cc}\ce{O}\phantom{..}\\ ||\phantom{..}\\\ce{H - C - OH} \end{array}\] |
| 2. | Acidic acid | C2H4O2 | \[\begin{array}{cc}\phantom{.}\ce{O}\\ \phantom{.}||\\\ce{CH3 - C - OH} \end{array}\] |
| 3. | Propionic acid | C3H6O2 | \[\begin{array}{cc}\phantom{........}\ce{O}\\ \phantom{........}||\\\ce{CH3 - CH2 - C - OH}\end{array}\] |
| 4. | n - Butyric acid | C4H8O2 | \[\begin{array}{cc}\phantom{...............}\ce{O}\\ \phantom{...............}||\\\ce{CH3 - CH2 - CH2 - C - OH}\end{array}\] |
| 5. | iso - Butyric acid | C4H8O2 | \[\begin{array}{cc}\phantom{.......}\ce{O}\\ \phantom{.......}||\\\ce{CH3 - CH - C - OH}\\ |\phantom{......}\\\ce{CH3}\phantom{...} \end{array}\] |
APPEARS IN
RELATED QUESTIONS
Structure of the compound whose IUPAC name is 5, 6 – dimethylhept - 2 - ene is
Describe the classification of organic compounds based on their structure.
Write the molecular formula of the first six members of homologous series of nitro – alkanes.
Which of the following is a hetero-aromatic benzenoid compound?
Which of the following yields only one type of monosubstituted chloroalkane upon chlorination?
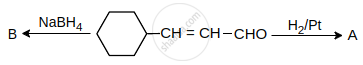
A and B are:
Identify primary, secondary, tertiary and quaternary carbon in the following compounds.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{.....}|\phantom{...............}\\
\ce{CH3}\phantom{..}\ce{CH3}\phantom{.............}
\end{array}\]
Identify primary, secondary, tertiary, and quaternary carbons in the following compound.
\[\begin{array}{cc}
\ce{CH3\phantom{.................}}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{....}|\phantom{................}\\
\ce{CH3 CH3\phantom{.............}}
\end{array}\]
Identify primary, secondary, tertiary and quaternary carbon in the following compound.
\[\begin{array}{cc}
\ce{CH3}\phantom{..................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{....}|\phantom{................}\\
\ce{CH3}\phantom{.}\ce{CH3}\phantom{..............}
\end{array}\]
Identify primary, secondary, tertiary and quaternary carbon in the following compound.
\[\begin{array}{cc}
\ce{CH3}\phantom{..................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{....}|\phantom{................}\\
\ce{CH3}\phantom{.}\ce{CH3}\phantom{..............}
\end{array}\]
