Advertisements
Advertisements
Question
Write the formula, structure and basicity of the following oxoacid:
Pyrophosphoric acid
Very Short Answer
Advertisements
Solution
Formula: H4P2O7
Structure: 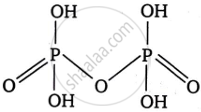
Basicity: Pyrophosphoric acid is tetrabasic because it has four ionisable hydrogen atoms attached to the four \[\ce{P - OH}\] groups, which can release protons (H+) in solution.
shaalaa.com
Is there an error in this question or solution?
