Advertisements
Advertisements
Question
Write the formula, structure and basicity of the following oxoacid:
Hypophosphorus acid
Very Short Answer
Advertisements
Solution
Formula: H3PO2
Structure:
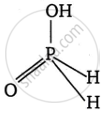
Basicity: It is a monobasic acid because only one acidic hydrogen (from the \[\ce{P - OH}\] group) is ionizable. The \[\ce{P - H}\] bonds do not ionize.
shaalaa.com
Is there an error in this question or solution?
