Advertisements
Advertisements
Question
Write balanced chemical equations for the action of hydrogen bromide on styrene in the presence of a peroxide
Answer in Brief
Advertisements
Solution
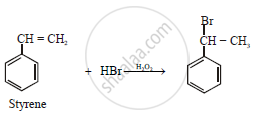
Balance equation is
\[\ce{\underset{\text{styrene}}{C6H5CH} = CH2 + HBr ->[Peroxide] C6H5CH2 - CH2Br }\]
shaalaa.com
Is there an error in this question or solution?
