Advertisements
Advertisements
Question
Write a short note on the following.
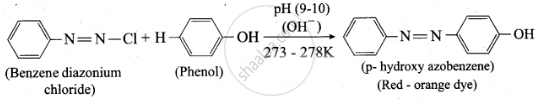
Coupling reaction
Short/Brief Note
Advertisements
Solution
Benzene diazonium chloride reacts with electron-rich aromatic compounds like phenol, aniline to form brightly coloured azo compounds. Coupling generally occurs at the para position. If para position is occupied then coupling occurs at the ortho position. Coupling tendency is enhanced if an electron-donating group is present at the para-position to \[\ce{-\overset{+}{N2}Cl^-}\] group. This is an electrophilic substitution.
shaalaa.com
Is there an error in this question or solution?
Chapter 13: Organic Nitrogen Compounds - Evaluation [Page 234]
