Advertisements
Advertisements
Question
Why are Zn2+ ions colourless while Ni2+ ions are green in colour?
Short/Brief Note
Advertisements
Solution
Zn2+ ions are colourless due to the absence of any unpaired electrons in its 3d-orbital.
Whereas, Ni2+ ions are green in colour due to the presence of 2 unpaired electrons d-d transitions are possible.
Zn2+ = 3d10 4s0
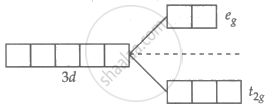
No unpaired electron in Zn2+.
∴ No d-d transition, colourless.
Ni2+ = 3d8 4s0
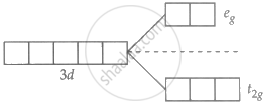
Unpaired electron present in Ni2+.
∴ d-d transition possible and are coloured.
shaalaa.com
Chemistry of Transition and Inner-transition Elements: D-block: 3d, 4d and 5d Series - D-block: 3d, 4d and 5d Series
Is there an error in this question or solution?
