Advertisements
Advertisements
Question
Which one of the following reactions does NOT give benzene as the product?
Options
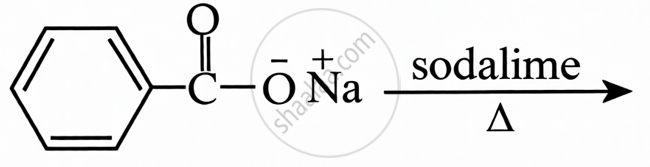

\[\ce{H - C ≡ C - H ->[red hot iron tube][at 873 K]}\]
MCQ
Advertisements
Solution

Explanation:
Sodium Benzoate + Sodalime: This is a decarboxylation reaction. Heating sodium benzoate with sodalime removes the carboxyl group to produce benzene.
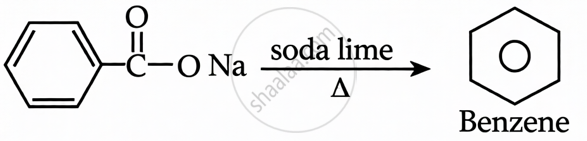
n-hexane + Mo2O3(773 K, 10-20 atm.): This is catalytic reforming (aromatization). n-hexane undergoes cyclization and dehydrogenation to form benzene.
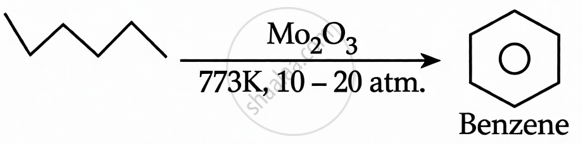
Ethyne + Red hot iron tube (873 K): This is the cyclic trimerization of ethyne (acetylene). Three molecules of ethyne combine to form benzene.

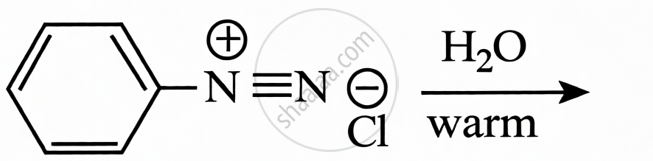
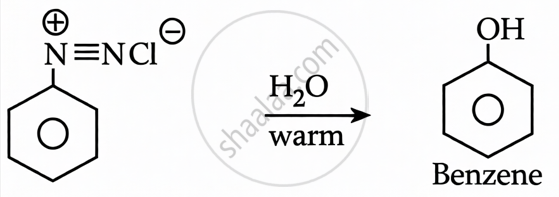
Benzene Diazonium Chloride + H2O (warm): When benzene diazonium chloride is warmed with water, it undergoes hydrolysis to produce phenol (C6H5OH), nitrogen gas, and HCl.
shaalaa.com
Is there an error in this question or solution?
