Advertisements
Advertisements
Question
Which one of the following does not have sp3 hybridisation?
Options
CH4
XeF4
H2O
NH3
MCQ
Advertisements
Solution
XeF4
Explanation:
| Molecular formula | Geometry | Bond and hybridisation |
| CH4 | \[\begin{array}{cc} \ce{H}\phantom{..}\ce{H} \phantom{..}\ce{H}\\\backslash\phantom{..}| \phantom{..}/\\\ce{C}\\|\\\ce{H}\end{array}\] |
4σ bond sp3 hybridisation |
| XeF4 | 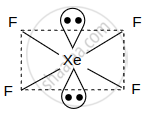 |
4σ bond +2 lone pairs sp3 d2 hybridisation |
| H2O | 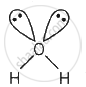 |
2σ bond +2 lone pairs sp3 hybridisation |
| NH3 | 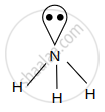 |
3σ bond + 1 lone pair sp3 hybridisation |
shaalaa.com
Hybridization
Is there an error in this question or solution?
