Advertisements
Advertisements
Question
Which one of the following compounds can exist as cis-trans isomers?
Options
Pent-1-ene
2-Methylhex-2-ene
1,1-Dimethylcyclopropane
1,2-Dimethylcyclohexane
Advertisements
Solution
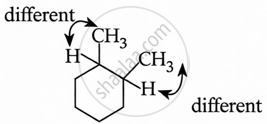
1,2-Dimethylcyclohexane
Explanation:
Cis and trans are geometric isomers. In order for geometric isomers to exist in alkenes, each doubly bonded carbon must be attached to a different group or atom.
In order for geometric isomers to exist in a cycloalkane, at least two sp3 carbons of the ring must be attached to different groups or atoms.
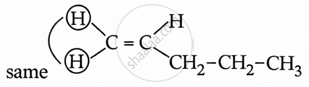
Pent-1-ene does not contain cis and trans isomers due to the presence of a single H atom on a doubly bound carbon atom.
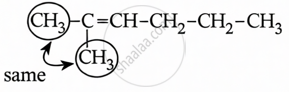
2-methylhex-2-ene does not contain geometric isomers due to the presence of two methyl groups on the same doubly bound carbon atom.
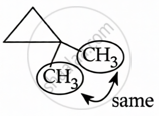
1,1-dimethylcyclopropane contains two identical methyl groups on each carbon atom.
1,2-dimethylcyclohexane exhibits geometric isomerism.