Advertisements
Advertisements
Question
Which one of the following alcohols is the strongest acid?
Options
Phenol
Methanol
Ethanol
t-butyl alcohol
MCQ
Advertisements
Solution
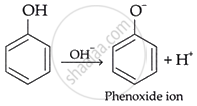
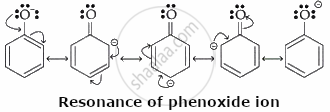
Phenol
Explanation:
Phenol is the most potent acid owing to the resonance stabilization of the phenoxide ion (C6H5O−). Upon the deprotonation of phenol, the negative charge on the oxygen atom is delocalized across the aromatic ring via resonance, enhancing its stability and elevating its acidity.
shaalaa.com
Is there an error in this question or solution?
2024-2025 (March) Official Board
