Advertisements
Advertisements
Question
Which one is most reactive towards SN1 reaction?
Options
C6H5CH(C6H5)Br
C6H5CH(CH3)Br
C6H5C(CH3)(C6H5)Br
C6H5CH2Br
MCQ
Advertisements
Solution
C6H5C(CH3)(C6H5)Br
Explanation:
As we know that SN1 reaction proceeds via formation of a carbocation intermediate.
Thus, the more stable the carbocation, the more reactive the aryl halide is toward the SN1 reaction.
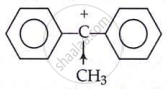
Here in this compound, +I effect of −CH3 and delocalisation of +ve charge over the two phenyl rings reduce the magnitude of the positive charge on the carbon atom, making it the most stable carbocation.
Hence,
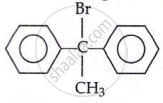 will undergo SN1 faster.
will undergo SN1 faster.
shaalaa.com
Is there an error in this question or solution?
