Advertisements
Advertisements
Question
Which of the following molecules is a polar molecule?
Options
MCQ
Advertisements
Solution
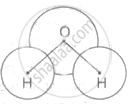
Explanation:
A polar molecule is one that has polar bonds and has a dipole moment that is greater than zero.
The bonds in H2O are inclined at an angle, resulting in a non-zero net dipole moment. As a result, H2O is a polar molecule, and the dipole moment is balanced in all other circumstances, therefore the net moment is zero.
shaalaa.com
Is there an error in this question or solution?