Advertisements
Advertisements
Question
Which of the following is INCORRECT for the following reaction?
\[\ce{2Zn_{(s)} + O2_{(g)} -> 2ZnO_{(s)}}\]
Options
Zn is the reducing agent.
O2 is the oxidizing agent.
Zn accepts electrons.
The oxidation number of O decreases from 0 to −2.
MCQ
Advertisements
Solution
Zn accepts electrons.
Explanation:
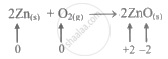
Zn is oxidation number rises from 0 to +2 when electrons are lost, making it a reducing agent that is also oxidised.
By gaining electrons, the oxidation number of O lowers from 0 to −2, indicating that O is an oxidising agent that is also reduced.
shaalaa.com
Is there an error in this question or solution?
