Advertisements
Advertisements
Question
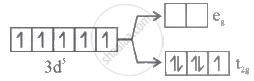
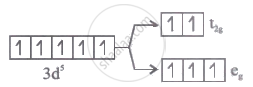
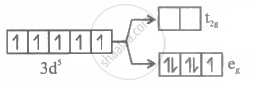
Which of the following energy level diagram for [FeF6]3- is correct on the basis of crystal field theory?
Options
MCQ
Advertisements
Solution
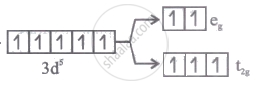
Explanation:
[FeF6]3- ⇒ x - 6 = - 3 Fe3+ = 
x = - 3 + 6
x = + 3
Fluorine is a weak ligand, .·. pairing does not occur.
shaalaa.com
Bonding in Coordination Compounds
Is there an error in this question or solution?