Advertisements
Advertisements
Question
Which gas is usually liberated when an acid reacts with a metal? Illustrate with an example. How will you test for the presence of this gas?
Answer in Brief
Advertisements
Solution
Hydrogen gas is usually liberated when an acid reacts with a metal.
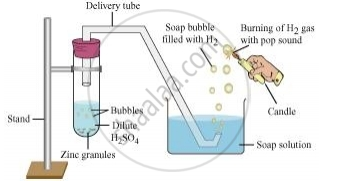
Take few pieces of zinc granules and add 5 ml of dilute H2SO4. Shake it and pass the gas produced into a soap solution. The bubbles of the soap solution are formed. These soap bubbles contain hydrogen gas.
H2SO4+Zn → ZnSO4 + H2 ↑
We can test the evolved hydrogen gas by its burning with a pop sound when a candle is brought near the soap bubbles.
shaalaa.com
Is there an error in this question or solution?
