Advertisements
Advertisements
Question
|
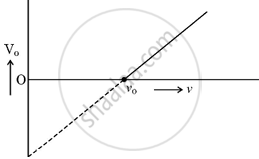
When a photon of suitable frequency is incident on a metal surface, photoelectron is emitted from it. If the frequency is below a threshold frequency (vo) for the surface, no photoelectron is emitted. For a photon of frequency v(v > vo), the kinetic energy of the emitted photoelectrons is h(v − vo). The photocurrent can be stopped by applying a potential Vo called ‘stopping potential’ on the anode. Thus maximum kinetic energy of photoelectrons Km = eVo = h(v − vo). The experimental graph between Vo and v for a metal is shown in figure. This is a straight line of slope m.
|
(i) The straight line graphs obtained for two metals ______.
- coincide each other.
- are parallel to each other.
- are not parallel to each other and cross at a point on v-axis.
- are not parallel to each other and do not cross at a point on v-axis.
(ii) The value of Planck’s constant for this metal is ______.
- `e/m`
- `1/(me)`
- me
- `m/e`
(iii) The intercepts on v-axis and Vo-axis of the graph are respectively ______.
- vo, `(hv_o)/e`
- vo, hvo
- `(hv_o)/e`, vo
- hvo, vo
OR
(iii) When the wavelength of a photon is doubled, how many times its wave number and frequency become, respectively?
- 2, `1/2`
- `1/2, 1/2`
- `1/2`, 2
- 2, 2
(iv) The momentum of a photon is 5.0 × 10−29 kg. m/s. Ignoring relativistic effects (if any), the wavelength of the photon is ______.
- 1.33 μm
- 3.3 μm
- 16.6 μm
- 13.3 μm
Advertisements
Solution
(i) The straight line graphs obtained for two metals are parallel to each other.
Explanation:
If two metals have different work functions, their straight line graphs are parallel to each other
(ii) The value of Planck’s constant for this metal is me.
Explanation:
From the graph:
Slope (m) = `h/e`
Then, h = e.m
(iii) The intercepts on v-axis and Vo-axis of the graph are respectively `bbunderline(v_o, (hv_o)/e)`.
Explanation:
From the graph, the intercept on the v-axis is vo, and the intercept on the vo-axis is hvo/e.
So, `(v_o, (hv_o)/e)`
OR
`bb(1/2, 1/2)`
Explanation:
Wave number (k) i = `(2 pi)/lambda`
When λ is doubled, k becomes half.
Frequency (f) = `c/lambda`
When λ is doubled, f becomes half.
So, `(1/2, 1/2)`
(iv) The momentum of a photon is 5.0 × 10−29 kg. m/s. Ignoring relativistic effects (if any), the wavelength of the photon is 13.3 μm.
Explanation:
Given: ρ = `h/lambda`
λ = `h/p`
= `(6.63 xx 10^-34)/(5.0 xx 10^-29)`
= 1.33 × 10−5 m
= 1.33 μm