Advertisements
Advertisements
Question
What types of halides are formed by group 15 elements? Discuss their structures and important characteristics.
Advertisements
Solution
1) Trihalides: When taken in excess, the elements of group 15 react directly with halogens to create MX3 trihalides. Except for NI3, all of the trihalides of N, P, As, Sb, and Bi are known. The following are some of these trihalides important characteristics.
(i) In the gaseous state, the trihalides (except from BiF3) have a pyramidal structure and are primarily covalent. As shown in the figure for PX3, the central group 15 element that is present in them is in a state of sp3 hybridisation and carries a single pair of electrons.
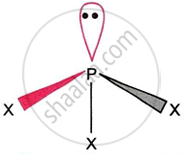
(ii) Water hydrolyses the trihalides with ease, with the exception of NF3 and BiF3. For other halides, however, the resulting compounds can change. For example,
\[\ce{NCl + 3H2O -> NH3 + \underset{acid}{\underset{Hypochlorous}{3HOCl}}}\]
\[\ce{PCl3 + 3H2O -> \underset{Phosphorus acid}{H3PO3} + 3HCl}\]
\[\ce{4AsCl3 + 6H2O -> As4O6 + 12HCl}\]
\[\ce{SbCl3 + H2O -> \underset{Antimony oxychloride}{SbOCl} + HCl}\]
\[\ce{BiCl3 + H2O -> \underset{Bismuth oxychloride}{BiOCl} + HCl}\]
(iii) The trihalides of P, As and Sb (specially fluorides) act as Lewis acids because they can combine with Lewis bases. For example,
\[\ce{PF3 + F2 -> PF5}\]
\[\ce{SbF3 + 2F- -> [SbF5]^{2-}}\]
2) Pentahalides: The first member of the group, nitrogen, is unable to expand its octet because its valence shell lacks d-orbitals, which prevents it from forming pentahalides. A few pentahalides of the type MX5 are formed by the remaining group members.
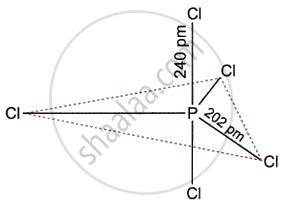
Phosphorus pentachloride (PCl5) is the most significant pentahalide produced by group 15 elements. As shown in the figure, it has a trigonal bipyramidal structure. It contains a phosphorus atom in the middle that is sp3d-hybridized. When heated, PCl5 converts into PCl3, which is thermally less stable than PCl3.
Phosphorus pentachloride is used as an industrial intermediate for the preparation of various phosphorus compounds.
