Advertisements
Advertisements
Question
What is the oxidation state of S in the following oxoacid?
Peroxodisulphuric acid
Very Short Answer
Advertisements
Solution
Peroxodisulphuric acid (H2S2O8)
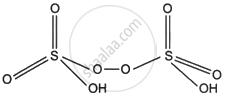
2 × (+1) + 2x + 6 × (−2) + 2 × (−1) = 0
2 + 2x − 12 − 2 = 0
2x − 12 = 0
⇒ 2x = 12
⇒ x = +6
Hence, the oxidation state of each ‘S’ in H2S2O8 is +6.
shaalaa.com
Is there an error in this question or solution?
