Advertisements
Advertisements
Questions
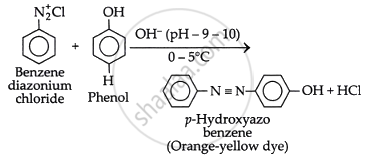
What is the action of benzene diazonium chloride on phenol in alkaline medium?
Give a balanced chemical equation for the following reaction:
Phenol is treated with ice cold alkaline solution of benzene diazonium chloride.
Write the reaction between benzene diazonium chloride and phenol in an alkaline medium.
Advertisements
Solution 1
The reaction of benzenediazonium chloride with phenol in a mild alkaline medium yields p-hydroxyazobenzene (orange dye).

Solution 2
Phenol is treated with ice-cold alkaline solution of benzene diazonium chloride to give an orange-yellow Azo dye (N = N linkage) compound.
APPEARS IN
RELATED QUESTIONS
Write the major products(s) in the following:

Write a note on Sandmeyer's reaction.
What is the action of benzene diazonium chloride on Aniline
An aromatic compound 'A' on heating with Br2 and KOH forms a compound 'B' of molecular formula C6H7N which on reacting with CHCl3 and alcoholic KOH produces a foul-smelling compound 'C'. Write the structures and IUPAC names of compounds A, B, and C.
A hydrocarbon ‘A’, \[\ce{(C4H8)}\] on reaction with \[\ce{HCl}\] gives a compound ‘B’, \[\ce{(C4H9Cl)}\], which on reaction with 1 mol of \[\ce{NH3}\] gives compound ‘C’, \[\ce{(C4H11N)}\]. On reacting with \[\ce{NaNO2}\] and \[\ce{HCl}\] followed by treatment with water, compound ‘C’ yields an optically active alcohol, ‘D’. Ozonolysis of ‘A’ gives 2 mols of acetaldehyde. Identify compounds ‘A’ to ‘D’. Explain the reactions involved.
A compound with the molecular formula C5H5N and having 3 double bonds will be
Diazo-coupling is useful to prepare ______.
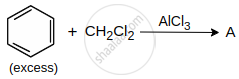
A is:
Write a structure of 2-Iodo-3 - methylpentane.
