Advertisements
Advertisements
Question
What is meant by bidentate ligands? Illustrate with examples.
Very Long Answer
Advertisements
Solution
The ligands which possess two donor atoms and can form two coordinate bonds with the central metal atom or ion are called bidentate ligands. Examples of bidentate ligands:
- Ethylenediamine (en): Ethylenediamine is a common bidentate ligand with two nitrogen atoms (each with a lone pair of electrons) that can coordinate to the metal ion.
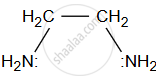
In the complex [Cu(en)2]2+ ethylenediamine acts as a bidentate ligand by coordinating to the copper ion via the two nitrogen atoms. - Glycine (NH2CH2COOH): Glycine is an amino acid that can act as a bidentate ligand due to the lone pairs of electrons on both the nitrogen (amino group) and oxygen (carboxyl group).
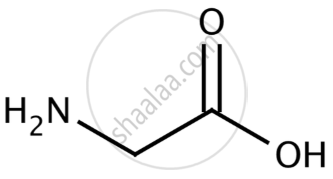
In the complex [Cu(gly)2]2+ glycine acts as a bidentate ligand by coordinating to the copper ion via both the nitrogen and oxygen atoms.
shaalaa.com
Is there an error in this question or solution?
